Anemia refers to a clinical condition where the concentration of hemoglobin (Hb), red blood cell count (RBC), and hematocrit (HCT) in a unit volume of circulating blood fall below the lower limits of the reference values for populations of the same age, gender, and region. It is one of the most common clinical symptoms. Clinically, hemoglobin (Hb) concentration is often used as a substitute for its diagnosis.
Generally, the diagnostic criteria are based on the standards established by the World Health Organization (WHO) in 1972. At sea level, anemia can be diagnosed when venous hemoglobin levels are below the following thresholds: 110 g/L for children aged 6 months to <6 years, 120 g/L for children aged 6–14 years, 130 g/L for adult males, 120 g/L for adult non-pregnant females, and 110 g/L for pregnant females. It should be noted that hemoglobin concentrations are naturally lower in infants, children, and pregnant women, while individuals living at high altitudes tend to have higher normal Hb values compared to those at sea level. Additionally, plasma volume increases during pregnancy, hypoproteinemia, congestive heart failure, splenomegaly, and macroglobulinemia. Under these conditions, even if the red blood cell count remains normal, anemia may appear to be present due to hemodilution and a reduced Hb concentration, potentially leading to a misdiagnosis. Conversely, in cases of dehydration or blood loss, where the volume of circulating blood decreases, hemoconcentration may cause Hb levels to appear elevated. This could lead to a missed anemia diagnosis, even when there is a reduction in red blood cells. Therefore, when determining the presence of anemia, these influencing factors should be carefully evaluated.
Classification
Anemia can be categorized according to different clinical characteristics. For instance, classification based on the progression speed includes acute and chronic anemia; classification by red blood cell morphology includes macrocytic anemia, normocytic anemia, and microcytic hypochromic anemia; classification by Hb concentration includes mild, moderate, severe, and very severe anemia; classification by bone marrow erythroid cell proliferation includes hypoproliferative anemia (such as aplastic anemia) and proliferative anemia (excluding aplastic anemia). While these classifications are useful for aiding diagnosis and guiding treatment, the following classification based on pathogenesis and/or etiology better reflects the fundamental pathology of anemia.
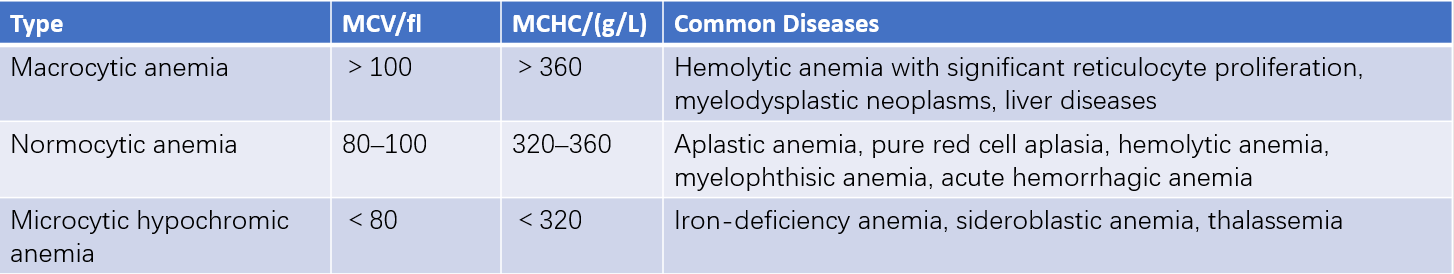
Table 1 Cytological classification of anemia
Note: MCV = Mean Corpuscular Volume; MCHC = Mean Corpuscular Hemoglobin Concentration
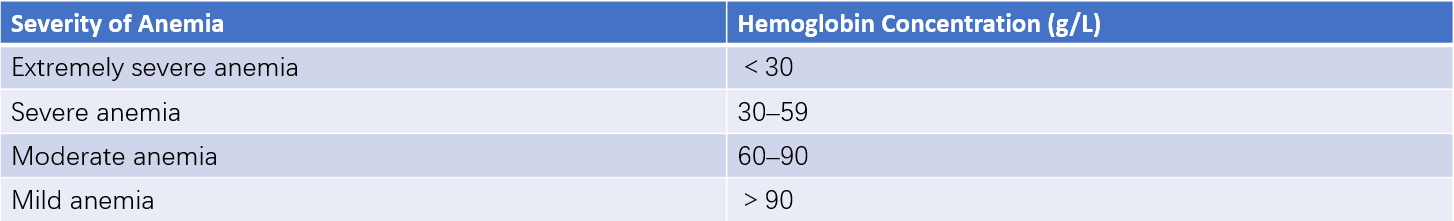
Table 2 Criteria for assessing the severity of anemia
Anemia Due to Decreased Red Blood Cell Production
Red blood cell production mainly depends on three factors: hematopoietic cells, hematopoiesis regulation, and hematopoietic raw materials.
- Hematopoietic Cells: These include pluripotent hematopoietic stem cells, myeloid progenitor cells, and various stages of erythroid precursors.
- Hematopoiesis Regulation: This involves both cellular regulation and factor-based regulation. Cellular regulation includes effects from bone marrow stromal cells, lymphocytes, and apoptosis (programmed cell death) of hematopoietic cells. Factor-based regulation includes positive and negative regulatory factors such as stem cell factor (SCF), interleukins (IL), granulocyte-macrophage colony-stimulating factor (GM-CSF), granulocyte colony-stimulating factor (G-CSF), erythropoietin (EPO), thrombopoietin (TPO), transforming growth factor (TGF), tumor necrosis factor (TNF), and interferons (IFN).
- Hematopoietic Raw Materials: These are the essential substances required for the proliferation, differentiation, metabolism, and structural assembly of hematopoietic cells, including proteins, lipids, vitamins (e.g., folic acid, vitamin B12), and trace elements (e.g., iron, copper, zinc). Abnormalities in any of these factors can result in decreased red blood cell production, ultimately causing anemia.
Anemia Caused by Hematopoietic Stem/Progenitor Cell Abnormalities
Aplastic Anemia (AA)
The development of AA is associated with primary and secondary defects in hematopoietic stem/progenitor cells. It is a type of anemia characterized by bone marrow failure in erythropoiesis.
Pure Red Cell Aplasia (PRCA)
PRCA is defined as anemia caused by the failure of the erythroid lineage in the bone marrow. It can be divided into congenital and acquired types. Congenital PRCA, also known as Diamond-Blackfan syndrome, is hereditary. Acquired PRCA includes primary and secondary subtypes. Since the 1970s, researchers have found that some cases of primary PRCA involve autoantibodies against erythropoietin (EPO) or proerythroblasts in the patient’s serum. Secondary PRCA is commonly associated with drug reactions, infections (bacterial or viral, such as parvovirus B19 or hepatitis viruses), autoimmune diseases, lymphoproliferative disorders (e.g., thymomas, lymphomas, plasma cell disorders, or chronic lymphocytic leukemia), certain myeloid malignant clone diseases (e.g., preleukemia states), and aplastic anemia crises. Based on disease progression and patient age, PRCA can be categorized into acute PRCA, chronic childhood PRCA (congenital), and chronic adult PRCA.
Congenital Dyserythropoietic Anemia (CDA)
CDA encompasses a group of refractory anemias caused by hereditary benign clonal abnormalities in erythroid stem/progenitor cells, characterized by ineffective erythropoiesis and morphological abnormalities of erythroid cells. Based on inheritance patterns, this condition is classified into autosomal recessive types and autosomal dominant types.
Malignant Clonal Disorders of the Hematopoietic System
This includes myelodysplastic neoplasms and various hematologic malignancies. These disorders are associated with qualitative abnormalities in pluripotent hematopoietic stem cells or myeloid stem/progenitor cells, leading to hyperproliferation, impaired differentiation, and disturbances in hematopoiesis regulation, ultimately resulting in insufficient production of mature red blood cells and anemia.
Anemias Caused by Abnormal Hematopoiesis Regulation
Anemia Due to Bone Marrow Stromal Cell Damage
Bone marrow necrosis, bone marrow fibrosis, bone sclerosis, osteopetrosis, bone marrow metastases of various extramedullary neoplastic disorders, and bone marrow infections or noninfectious inflammations can all damage bone marrow stromal cells and the hematopoietic microenvironment (and potentially hematopoietic cells themselves), impairing blood cell production and resulting in anemia.
Anemia Due to Hyperfunction of Lymphocytes
T-cell hyperfunction may lead to hematopoietic failure through direct cytotoxic killing of hematopoietic cells (e.g., via perforins by cytotoxic T-cells) and/or T-cell factor-mediated apoptosis of hematopoietic cells (as seen in aplastic anemia, AA). B-cell hyperfunction may result in the production of bone marrow autoantibodies, which disrupt or suppress hematopoietic cells, ultimately impairing hematopoiesis and leading to immune-related pancytopenia.
Anemia Due to Abnormal Levels of Hematopoietic Regulatory Factors
Conditions such as renal insufficiency, pituitary or thyroid hypofunction, and liver diseases may cause reduced production of erythropoietin (EPO), resulting in anemia. Tumorous diseases or certain viral infections can induce excessive production of negative hematopoietic regulatory factors, including tumor necrosis factor (TNF), interferons (IFN), and inflammatory cytokines, which suppress hematopoiesis and contribute to anemia. In recent years, hepcidin, a key hormone that regulates dietary iron absorption and iron release from macrophages, has been found to play a significant role. During anemia and hypoxia, hepcidin secretion decreases, promoting iron utilization by red blood cells. However, infections and pro-inflammatory cytokines stimulate hepcidin secretion, reducing plasma free iron levels and leading to impaired iron utilization. Anemia of chronic disease falls into this category.
Anemia Due to Enhanced Hematopoietic Cell Apoptosis
Some researchers have proposed a "dual pathogenesis" theory for paroxysmal nocturnal hemoglobinuria (PNH), where PIGA gene mutations confer intrinsic anti-apoptotic properties to PNH clone cells, leading to clonal expansion of abnormal hematopoietic stem cells, while T-cell-mediated apoptosis occurs in normal hematopoietic cells. The failure of myeloid hematopoiesis in AA is also largely attributed to increased apoptosis.
Anemia Caused by Deficient or Impaired Utilization of Hematopoietic Raw Materials
Anemia from Folate or Vitamin B12 Deficiency or Utilization Impairment
This type of anemia, commonly referred to as megaloblastic anemia in clinical settings, arises from either absolute or relative deficiencies of folate or vitamin B12 or their impaired utilization, due to various physiological or pathological factors.
Iron Deficiency and Impaired Iron Utilization Anemia
This is the most common form of anemia in clinical practice. Deficiencies or impaired utilization of iron affect hemoglobin synthesis, leading some researchers to classify this as a disorder of heme synthesis. Morphologically, red blood cells in this type of anemia appear smaller with an enlarged central pallor, typically presenting as microcytic hypochromic anemia.
Anemias Caused by Excessive Red Blood Cell Destruction
This category includes hemolytic anemias (HA), characterized by increased red blood cell destruction.
Anemias Caused by Blood Loss
Blood loss anemia can be classified based on the rate of blood loss (acute or chronic), the volume of blood loss (mild, moderate, or severe), and the underlying etiology. Etiologies may include hemorrhagic or coagulation disorders (e.g., immune thrombocytopenic purpura, hemophilia, severe liver disease) or non-hemorrhagic and non-coagulation-related conditions (e.g., trauma, tumors, tuberculosis, bronchiectasis, peptic ulcers, liver disease, hemorrhoids, and genitourinary diseases). Chronic blood loss anemia is often accompanied by iron deficiency anemia.
Clinical Manifestations
The most common systemic symptom of anemia is fatigue. Its clinical presentation is related to five primary factors: the underlying cause of anemia (including associated conditions leading to anemia), the degree of reduction in the blood's oxygen-carrying capacity caused by anemia, the extent of decreased blood volume, the rate at which anemia develops, and the compensatory and adaptive responses of systems such as circulatory, respiratory, and hematologic to the condition. The main clinical manifestations of anemia are as follows.
Nervous System
Common symptoms of anemia include headache, dizziness, lethargy, fainting, insomnia, dreams, tinnitus, blurred vision, memory decline, and difficulty concentrating. Some of these symptoms are due to hypoxia in brain tissue caused by anemia, while others are attributable to acute blood loss anemia leading to insufficient blood volume or lower blood pressure. Severe hemolysis may result in these symptoms through hyperbilirubinemia or elevated free hemoglobin levels. Primary diseases causing anemia, such as leukemic infiltration into the central nervous system, may also contribute to neurological symptoms, as may complications like intracranial or retinal hemorrhage seen in disorders such as aplastic anemia. Peripheral neuropathy, such as numbness in extremities, may occur as a complication of anemia, particularly in cases of vitamin B12 deficiency-related megaloblastic anemia. In children with iron-deficiency anemia, symptoms may include irritability, restlessness, or even impaired cognitive development.
Skin and Mucosa
Pallor of the skin and mucous membranes is the primary manifestation of anemia in these tissues, primarily caused by the redistribution of effective blood volume through neurohumoral regulation. Blood supply is prioritized to vital organs (e.g., brain, heart, kidneys, liver, and lungs) at the expense of relatively less essential structures (e.g., skin and mucous membranes). Additionally, reduced red blood cell and hemoglobin content per unit volume of blood contributes to the paler appearance. Rough or lusterless skin, ulcers, or other damage may also manifest due to reduced blood supply, nutritional deficiencies, or primary conditions associated with anemia (e.g., folic acid, vitamin B12, or iron deficiency, as well as autoimmune diseases). Hemolytic anemia, particularly extravascular hemolysis, may result in skin and mucosal jaundice. Certain hematopoietic malignancies associated with anemia may present with skin abnormalities (e.g., chloromas).
Respiratory System
In mild anemia, the body’s compensatory mechanisms may maintain resting respiratory rates within the normal range. However, physical activity creates a state of hypoxia and increased partial pressure of carbon dioxide, stimulating the respiratory centers and leading to more rapid and deeper breathing. In severe anemia, dyspnea or even orthopnea may occur even at rest. Additionally, complications of anemia and its underlying causes may affect the respiratory system. For example, aplastic anemia may be complicated by respiratory infections, leukemic anemia may involve infiltration into pulmonary tissue, anemic complications of systemic lupus erythematosus may result in "lupus lung," and chronic transfusions may lead to hemosiderosis of the lungs, presenting with corresponding pulmonary symptoms, signs, and radiographic findings.
Circulatory System
In acute blood loss anemia, the circulatory system's primary response is to the reduced blood volume, leading to peripheral vasoconstriction, tachycardia, and subjective sensations such as palpitations. In non-hemorrhagic anemia, blood volume is typically preserved, and circulatory changes primarily reflect the heart’s response to tissue hypoxia. In mild anemia, symptoms may only become apparent during physical activity, with tachycardia and palpitations. In moderate-to-severe anemia, these symptoms may occur even at rest, and the severity increases with greater anemia and physical activity, placing additional workload on the heart. Chronic anemia can lead to overworked cardiac function and insufficient blood supply, eventually resulting in anemia-induced heart disease. This condition may involve not only heart rate changes but also arrhythmias, structural abnormalities, and even heart failure. Repeated transfusions leading to "hemochromatosis" may contribute to cardiac dysfunction and changes in heart rate and rhythm. Additionally, primary diseases causing anemia that involve the heart or blood vessels may lead to cardiovascular changes.
Digestive System
Digestive system diseases that cause anemia may present with symptoms of the primary condition before or concomitant with anemia. Diseases outside the digestive system that cause anemia may also affect digestive function. Anemia itself can impact the digestive system, resulting in functional or structural changes such as reduced glandular secretions or glandular atrophy, leading to diminished digestive capabilities, indigestion, abdominal distension, reduced appetite, and alterations in bowel habits or stool characteristics. Chronic hemolysis may lead to gallstone formation and/or inflammation. Iron-deficiency anemia may cause symptoms such as dysphagia or a sensation of a lump in the throat. Iron-deficiency anemia caused by hookworm infections may present with pica. Megaloblastic anemia or pernicious anemia can result in glossitis, atrophy of lingual papillae, "beefy" tongues, or smooth, glossy tongues.
Urinary System
Renal anemia is often associated with clinical manifestations of the primary kidney disease both before and during the onset of anemia. Extravascular hemolysis may result in bilirubinuria and elevated urinary urobilinogen, while intravascular hemolysis may lead to the presence of free hemoglobin and hemosiderin in the urine. Severe cases may exhibit tubular occlusion by free hemoglobin, causing oliguria, anuria, or even acute renal failure. Acute, severe blood loss anemia may lead to decreased renal blood flow due to insufficient blood volume, resulting in oliguria or anuria. If prolonged, this condition may cause renal impairment.
Endocrine System
Anemia caused by massive hemorrhage during childbirth may result in pituitary ischemia and necrosis, leading to Sheehan's syndrome. Prolonged anemia may impair the functions of the thyroid gland, gonads, adrenal glands, and pancreas, and may alter the secretion of erythropoietin (EPO) and gastrointestinal hormones. Certain autoimmune conditions that affect the hematopoietic system may simultaneously involve one or more endocrine organs, resulting in hormonal imbalances or abnormal secretions.
Reproductive System
In males, chronic anemia can lead to ischemia and necrosis of the spermatogenic cells in the testes, affecting testosterone secretion and diminishing male secondary sexual characteristics. In females, anemia may influence the secretion of female hormones and also result in menorrhagia due to abnormalities in the quantity or quality of coagulation factors and platelets.
Immune System
Anemia secondary to immune system diseases is accompanied by the clinical manifestations of the primary immune-related conditions. Anemia itself can also induce changes in the immune system. A reduction in red blood cells may decrease their opsonization role in fighting infections caused by pathogenic microorganisms. A decrease in complement receptor 1 (CR1) on red blood cell membranes may impair non-specific immune functions. Repeated blood transfusions in anemic patients can affect T-cell subpopulations. Certain drugs used to treat anemia are also known to alter a patient's immune function.
Hematologic System
Changes in peripheral blood primarily manifest as alterations in blood cell counts, morphology, and biochemical composition. Some cases may also involve abnormalities in plasma or serum components. The most notable change in cell count is a reduction in red blood cells, accompanied by decreases in hemoglobin (Hb), hematocrit, and possibly reticulocyte counts. Abnormalities in white blood cell or platelet count (including leukocyte differentials) may also be present. Morphological changes in blood cells include variations such as macrocytic, microcytic, or normocytic anemia, as well as abnormal shapes observed in erythrocytes, leukocytes, and platelets.
Biochemical abnormalities in red blood cells generally fall into two categories: first, increased synthesis of 2,3-diphosphoglycerate (2,3-DPG) within red blood cells may occur, lowering the oxygen affinity of hemoglobin and shifting the oxygen dissociation curve to the right, thereby facilitating improved oxygen delivery to tissues; second, alterations related to the specific type of anemia, such as abnormalities in red blood cell membranes, enzymes, hemoglobin, or qualitative changes in leukocytes and platelets associated with certain types of anemia. Plasma or serum abnormalities are often seen in plasma cell disorder-related anemia (e.g., elevated M-proteins or changes in calcium and phosphorus levels), hemolytic anemia (e.g., increased free hemoglobin, decreased haptoglobin, elevated serum potassium, and heightened levels of either indirect or conjugated bilirubin), anemia with disseminated intravascular coagulation (e.g., abnormalities in various plasma coagulation factors and fibrinolysis components), anemia associated with liver disease and renal anemia (e.g., hypoproteinemia or accumulation of metabolic byproducts).
Changes in hematopoietic organs are primarily observed in the bone marrow. Different types of anemia present with varying levels of nucleated cell proliferation in the bone marrow. Additionally, diseases with differing causes or pathogenesis result in significant heterogeneity in the morphology, proportions, location, ultrastructure, histochemical reactions, antigen expression, chromosomal karyotypes, oncogene rearrangements or overexpression, and in vitro colony formation of hematopoietic stem/progenitor cells in granulocyte, erythroid, monocyte, megakaryocyte, and lymphocyte lineages. Anemia caused by hematologic malignancies often presents with hepatomegaly, splenomegaly, and lymphadenopathy. Hemolytic anemia may involve hepatomegaly or splenomegaly. Bone marrow fibrosis and anemia with hypersplenism are frequently associated with splenomegaly.
Diagnosis
A detailed inquiry into the present medical history, past medical history, family history, nutritional history, menstrual history, reproductive history, and history of exposure to risk factors is necessary. The present medical history provides information regarding the onset, progression, severity, complications, potential triggers, and response to intervention in anemia. Past medical history can offer clues about the underlying primary disease causing anemia. Family history aids in understanding any genetic predisposition to anemia. Nutritional, menstrual, and reproductive histories can assist in diagnosing anemias caused by deficiencies in hematopoietic materials such as iron, folic acid, or vitamin B12, as well as anemias related to blood loss. A history of exposure to risk factors such as radiation, chemical toxins or drugs, endemic regions, or pathogenic microorganisms is crucial for diagnosing anemia related to hematopoietic tissue damage or infections.
A comprehensive physical examination helps in assessing the following aspects:
- The systemic effects of anemia, such as the degree of pallor in the skin and mucous membranes, changes in heart rate or rhythm, and abnormalities in respiratory posture or frequency.
- Accompanying signs of anemia, including manifestations of hemolysis (e.g., jaundice of the skin, mucosa, or sclera, signs of biliary inflammation, hepatomegaly, or splenomegaly), bleeding (e.g., purpura or ecchymosis on the skin and mucous membranes, signs of hemorrhage in the fundus, central nervous system, genitourinary tract, or digestive tract), infiltration (e.g., skin chloromas, subcutaneous masses, lymphadenopathy, hepatomegaly, or splenomegaly), infections (e.g., fever, systemic reactions, or signs of infection foci), malnutrition (e.g., dry skin, mucosa, or hair, mucosal ulcers, atrophy of the lingual papillae, koilonychia, or deep sensory deficits in the nervous system), and autoimmune responses (e.g., skin or mucosal damage, joint involvement).
Laboratory tests for diagnosing anemia include complete blood count tests, bone marrow examinations, and tests for the underlying mechanisms of anemia.
Complete Blood Count Tests
These tests determine the presence of anemia and whether it is accompanied by changes in leukocyte or platelet counts. Red blood cell parameters (MCV, MCH, and MCHC) reflect changes in red blood cell size and hemoglobin content and provide clues for identifying the pathological mechanisms of anemia. Hemoglobin measurement assists in assessing the severity of anemia. Reticulocyte counts indirectly reflect red cell production in the bone marrow or compensatory responses to anemia. Peripheral blood smears allow the evaluation of changes in the number or morphology of red cells, white cells, and platelets, as well as the presence of abnormalities such as malaria parasites or atypical cells.
Bone Marrow Examinations
Bone marrow tests include bone marrow smear cytological classification and bone marrow biopsy. Cytological classification from smears reveals the degree of bone marrow proliferation, cellular composition, ratios, and morphological changes. Biopsy provides insights into the structure of hematopoietic tissue, proliferation levels, cellular composition, and morphology. Bone marrow examination can indicate whether hematopoietic function is normal or whether there are neoplastic changes, necrosis, fibrosis, osteosclerosis, or hypoplasia in hematopoietic tissues. When results conflict with blood routine tests, multiple-site bone marrow examinations should be performed.
Tests for Mechanisms of Anemia
These include assessments of iron metabolism and evaluation of primary diseases causing iron deficiency for iron-deficiency anemia; measurements of serum folate and vitamin B12 levels as well as identification of underlying diseases leading to these deficiencies in megaloblastic anemia; investigation of primary diseases causing blood loss in hemorrhagic anemia; and tests such as red cell lifespan determination (via CO breath tests), as well as evaluation of red cell membrane, enzymes, globin, heme, autoantibodies, alloantibodies, or PNH clones for hemolytic anemia. In aplastic anemia, tests are conducted to identify abnormalities in hematopoietic stem cells (e.g., chromosomal, antigen expression, cell cycle, function, or gene studies), T-cell regulation (e.g., T-cell subsets and cytokine production), B-cell regulation (e.g., marrow autoantibodies), and primary diseases causing hematopoietic malfunction or secondary anemia from hematopoietic malignancies or systemic conditions.
Clinical data obtained from medical history, physical examination, and laboratory tests typically clarifies the mechanisms or causes of anemia, enabling the diagnosis of the underlying condition.
Treatment
Treatment for anemia-related diseases involves symptomatic management and etiological treatment.
Symptomatic Treatment
The aim of symptomatic treatment is to mitigate the life-threatening effects of severe blood cell depletion, thereby providing time for etiological intervention. Specific measures include administering red blood cell transfusions to patients with severe anemia, the elderly, or those with cardiorespiratory insufficiency to correct anemia and alleviate hypoxia. Patients with acute massive hemorrhage may require immediate transfusion of whole blood, red blood cells, and plasma to restore blood volume and correct anemia. For anemia accompanied by bleeding, treatment strategies should address the specific mechanism of bleeding (e.g., platelet transfusion for severe thrombocytopenia). For anemia complicated by infections, appropriate anti-infective therapy may be necessary. Supportive treatments should be tailored to the affected organ and the degree of functional impairment in cases of anemia with multi-organ dysfunction. In patients with congenital hemolytic anemia complicated by hemochromatosis due to repeated transfusions, iron chelation therapy is indicated.
Etiological Treatment
Treating the underlying cause of anemia targets the disease mechanism. In iron-deficiency anemia, iron supplementation and treatment of the primary disease causing iron deficiency are required. In megaloblastic anemia, supplementation with folic acid or vitamin B12 is necessary. Hemolytic anemia may require glucocorticoids or splenectomy, with splenectomy showing definitive efficacy in hereditary spherocytosis. Hematopoietic stem cell transplantation is used in anemia caused by abnormal hematopoietic stem cells. In aplastic anemia, treatments such as anti-lymphocyte or anti-thymocyte globulins, cyclosporine, and positive hematopoietic regulators (e.g., androgens, G-CSF, GM-CSF, or EPO) are employed. For anemia related to chronic disease or renal dysfunction, EPO is the treatment of choice. Tumor-associated anemia may require chemotherapy or radiotherapy. Immunosuppressive agents are used for immune-related anemia. In secondary anemias, resolution of the primary disease is key to effective treatment.