Myelodysplastic neoplasms (MDS), also known as myelodysplastic syndrome, are a heterogeneous group of myeloid neoplastic diseases originating from hematopoietic stem cells, characterized by cytopenia and morphological dysplasia. These diseases can occur in both males and females at any age, with approximately 80% of cases observed in patients over 60 years old.
Etiology and Pathogenesis
The exact causes of primary MDS are still unclear. Secondary MDS has been observed in individuals with frequent exposure to alkylating agents, topoisomerase inhibitors, radiation, or organic toxins.
MDS is classified as a clonal disorder of hematopoietic stem cells. Abnormal clonal cells in the bone marrow exhibit differentiation and maturation defects, leading to dysplastic and ineffective hematopoiesis, with a high risk of progression to acute myeloid leukemia (AML). Some MDS patients may exhibit genetic mutations in hematopoietic cells, epigenetic alterations, chromosomal abnormalities, or abnormalities in the bone marrow microenvironment. These changes may play a role in the multifactorial, multistep, and dynamically progressive development of MDS.
Classification and Clinical Manifestations
The French-American-British (FAB) classification categorizes MDS into five types based on the proportion of blasts, morphological changes in the peripheral blood and bone marrow, and monocyte counts:
- Refractory anemia (RA),
- Refractory anemia with ringed sideroblasts (RAS/RARS),
- Refractory anemia with excess blasts (RAEB),
- Refractory anemia with excess blasts in transformation (RAEB-t),
- Chronic myelomonocytic leukemia (CMML).

Table 1 FAB classification of MDS
The 5th Edition of the WHO Classification of Tumors of Hematopoietic and Lymphoid Tissues emphasizes a genetic-based classification of MDS. It introduces two specific subtypes (MDS with low blasts [MDS-LB] and MDS with increased blasts [MDS-IB]) and eliminates the category "MDS, unclassifiable." The WHO also replaced the term "myelodysplastic syndrome" with "myelodysplastic neoplasm" to highlight the neoplastic nature of the disease. According to WHO criteria, 20% bone marrow blasts remain a diagnostic cutoff between MDS and AML, although MDS-IB2 may clinically be considered equivalent to AML in certain therapeutic contexts.
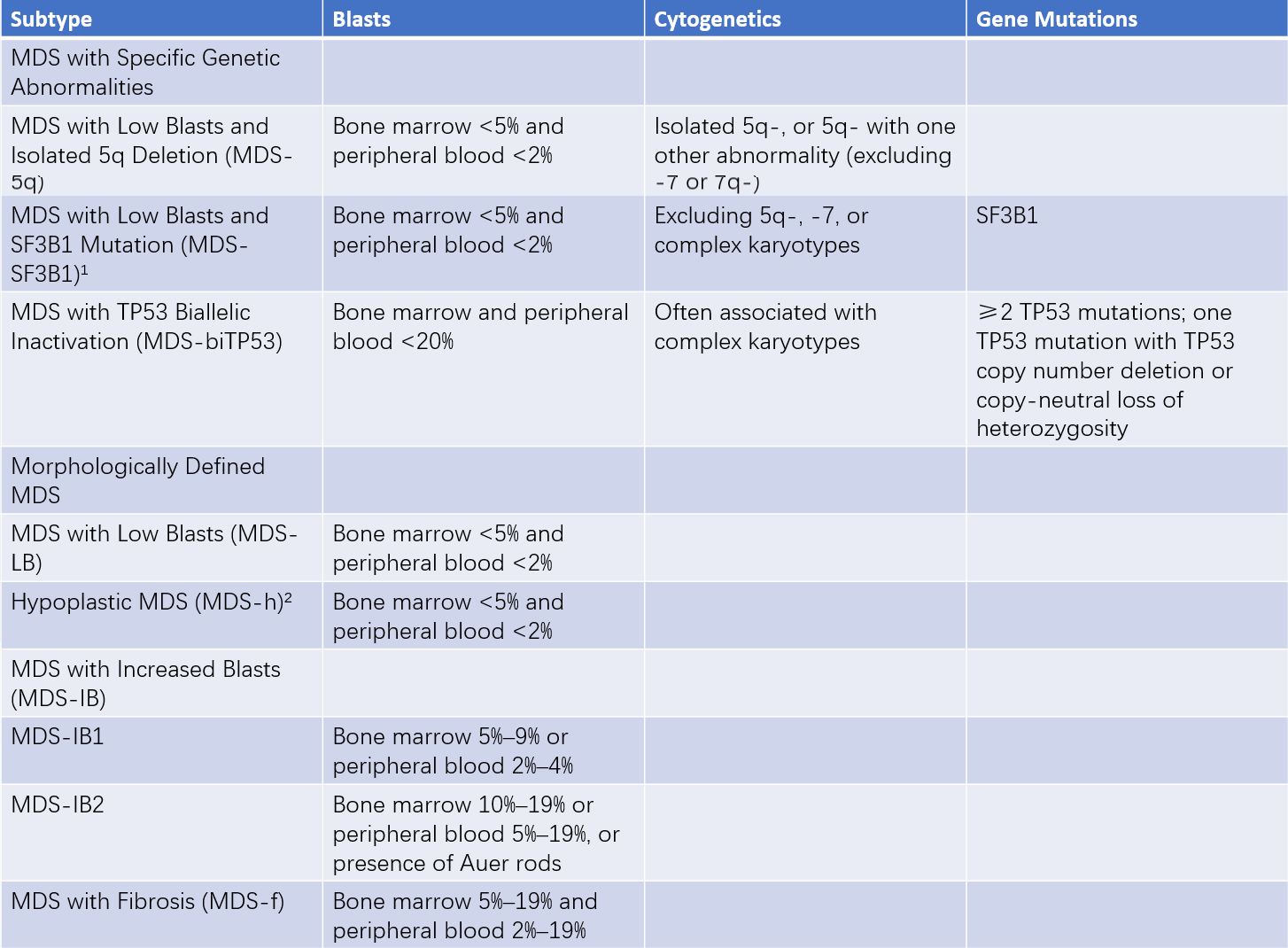
Table 2 WHO fifth edition classification of MDS
Notes:
1, Ringed sideroblasts ≥15% may replace SF3B1 mutations, labeled as "MDS with Low Blasts and Ringed Sideroblasts."
2, Hypoplastic MDS is defined as bone marrow cellularity ≤25% (age-adjusted).
Nearly all MDS patients experience anemia-related symptoms, such as fatigue and lethargy. About 60% of MDS patients exhibit neutropenia, accompanied by neutrophil dysfunction, which increases susceptibility to infections. Approximately 20% of MDS-related deaths are attributed to infections. Additionally, 40%–60% of patients present with thrombocytopenia, which can worsen progressively as the disease advances.
Laboratory Tests
Peripheral Blood and Bone Marrow Findings
Persistent reduction in one or more lineage of blood cells is commonly observed, with hemoglobin levels <100 g/L, neutrophil counts <1.8×109/L, and platelet counts <100×109/L. Bone marrow activity often shows hyperplasia, although some patients may exhibit hypocellular marrow.
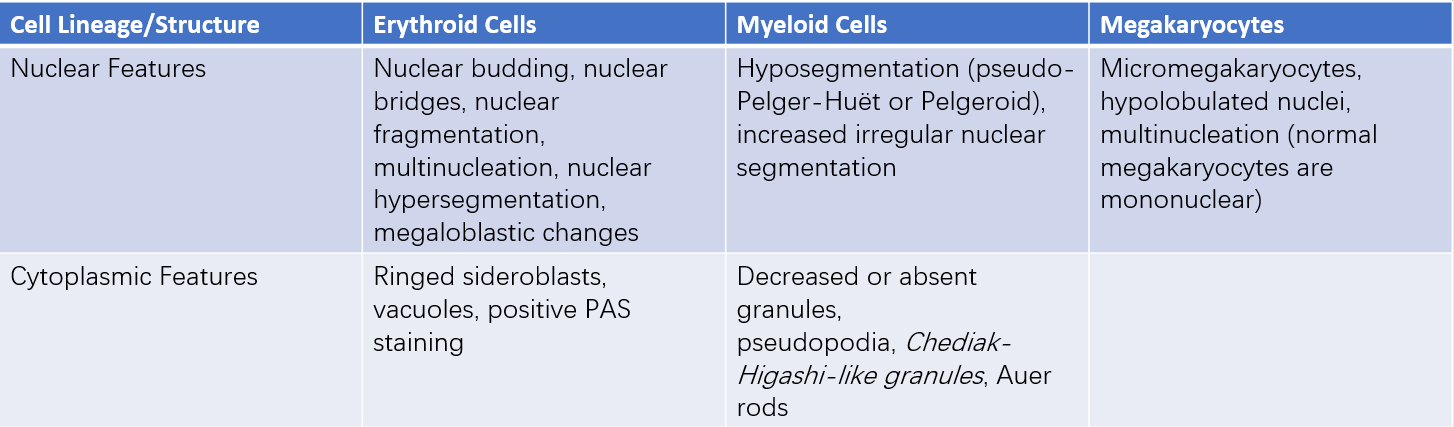
Table 3 Common morphological dysplasia in MDS
Cytogenetic Analysis
Clonal chromosomal abnormalities are observed in 40%–70% of MDS cases, predominantly involving deletions. The most frequent abnormalities include +8, -5/del(5q), -7/del(7q), and del(20q). Fluorescence in situ hybridization (FISH) techniques improve the detection rate of cytogenetic abnormalities.
Pathological Examination
Bone marrow biopsy provides critical information about the degree of cellular proliferation in the bone marrow, the number of megakaryocytes, the population of blasts, bone marrow fibrosis, and tumor metastasis to the bone marrow. This examination is essential for excluding other potential causes of cytopenia or related diseases.
Immunological Testing
Flow cytometry can identify phenotypic abnormalities in bone marrow cells of MDS patients. This method is valuable for distinguishing low-risk MDS from non-clonal conditions associated with cytopenia.
Molecular Biological Testing
Common gene mutations associated with MDS include TET2, RUNX1, ASXL1, DNMT3A, EZH2, and SF3B1. Mutation analysis has significant utility in the differential diagnosis of MDS and in risk stratification.
Diagnosis and Differential Diagnosis
The diagnosis of myelodysplastic syndromes (MDS) can be established based on cytopenia, corresponding clinical symptoms, morphological dysplasia, cytogenetic abnormalities, and pathological changes. Despite morphological dysplasia being characteristic of MDS, its presence alone does not confirm the diagnosis. There is no "gold standard" for diagnosing MDS, as it remains a diagnosis of exclusion. MDS often requires differentiation from the following conditions:
Chronic Aplastic Anemia (CAA)
CAA often needs to be differentiated from hypoplastic MDS. In hypoplastic MDS, reticulocyte levels can be normal or elevated, nucleated red blood cells may appear in peripheral blood, and bone marrow typically shows pronounced morphological dysplasia with normal or increased proportions of early-stage cells. Additionally, 40%–60% of MDS patients exhibit clonal cytogenetic abnormalities, whereas CAA generally lacks such abnormalities.
Paroxysmal Nocturnal Hemoglobinuria (PNH)
PNH can also present with pancytopenia and morphological dysplasia. However, PNH tests reveal the absence of anchor proteins on peripheral blood cell surfaces, a positive Ham test, and evidence of intravascular hemolysis.
Megaloblastic Anemia
Morphological dysplasia observed in MDS may include megaloblastic changes, which can mimic megaloblastic anemia. However, megaloblastic anemia results from folate or vitamin B12 deficiency, which is correctable by supplementation. In contrast, MDS does not involve deficiencies in folate or vitamin B12, and treatment with these supplements is ineffective.
Treatment and Prognosis
The natural course and prognosis of MDS vary significantly, emphasizing the importance of risk stratification in guiding treatment decisions. The Revised International Prognostic Scoring System (IPSS-R) evaluates prognosis based on the degree of cytopenia, the proportion of bone marrow blasts, and chromosomal abnormalities. It also serves as a framework for treatment planning. Recently, the International Working Group proposed incorporating 31 MDS-related gene mutations into the new IPSS-M prognostic model.
For patients with an IPSS-R score of ≤3.5, treatment primarily focuses on improving hematopoiesis and quality of life through supportive care, hematopoietic-stimulating therapies, hypomethylating agents, and biological response modifiers. For higher-risk groups, treatment is aimed at altering the disease trajectory using hypomethylating agents, chemotherapy, and hematopoietic stem cell transplantation (HSCT).
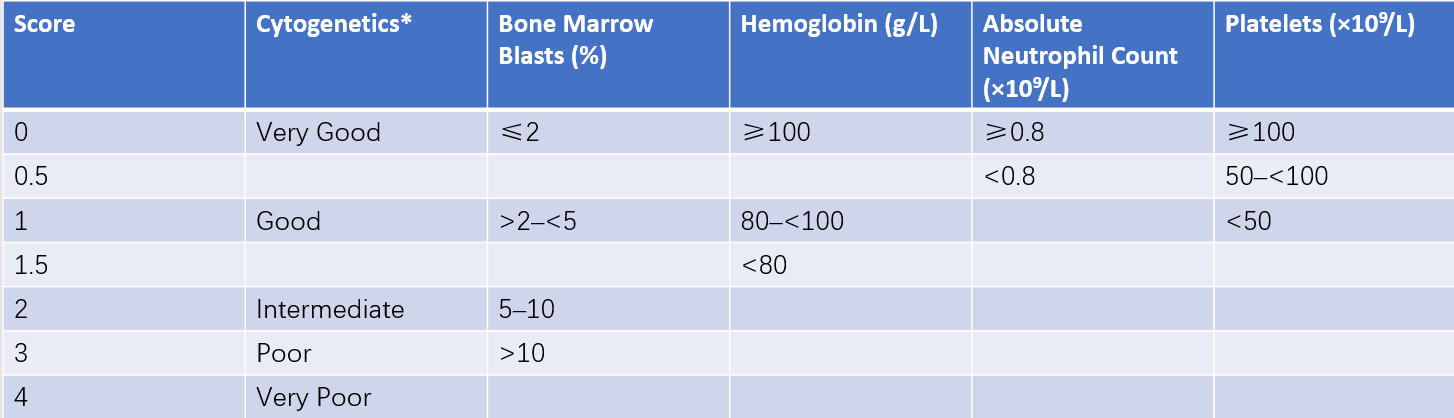
Table 4 Revised international prognostic scoring system for MDS (IPSS-R)
Notes:
Cytogenetics Categories:
Very Good: del(11q), -Y
Good: Normal karyotype, del(20q), del(12p), del(5q), or del(5q) with one additional abnormality
Intermediate: +8, del(7q), i(17q), +19, and other single or double independent chromosomal abnormalities
Poor: -7, inv(3)/t(3q)/del(3q), or -7/7q- with one additional abnormality; complex karyotype (3 abnormalities)
Very Poor: Complex karyotype (>3 abnormalities)
IPSS-R Risk Categories:
Very Low (VL): ≤1.5 points
Low (L): >1.5–≤3 points
Intermediate (Int): >3–≤4.5 points
High (H): >4.5–≤6 points
Very High (VH): >6 points
Supportive Care
Patients with severe anemia or bleeding symptoms can receive red blood cell and platelet transfusions. For those with neutropenia and deficiencies, infection prevention and management are vital. Iron chelation therapy is suitable for individuals experiencing iron overload due to prolonged transfusion.
Hematopoietic-Stimulating Therapy
Therapies such as erythropoietin (EPO) or androgens may improve hematopoietic function in some patients.
Biological Response Modifiers
Thalidomide and lenalidomide have demonstrated efficacy, particularly in MDS with isolated del(5q). Antithymocyte globulin (ATG) and/or ciclosporin may be used in a small subset of very low-risk MDS patients.
Hypomethylating Agents
Hypomethylating agents, such as azacitidine and decitabine, can reverse hypermethylation of tumor suppressor gene promoters, alter gene expression, reduce transfusion dependency, improve quality of life, and delay progression to AML. Approximately 90% of patients who respond to azacitidine treatment achieve responses within six cycles.
Combination Chemotherapy
For MDS patients with an IPSS-R score >3.5, AML induction regimens or preconditioning therapies can be employed. Preconditioning regimens are generally better tolerated than standard AML chemotherapy in elderly or physically frail patients. Preconditioning regimens may also be combined with hypomethylating agents.
Allogeneic Hematopoietic Stem Cell Transplantation (HSCT)
Currently, HSCT is the only potential curative treatment for MDS. Patients with an IPSS-R score >3.5 should be evaluated for eligibility for HSCT, particularly younger patients, those with increased blasts, or those with poor-prognosis cytogenetics. For lower-risk patients with transfusion dependency or unsuccessful hypomethylating treatment, HSCT may be considered after reducing iron overload.