Thyrotoxicosis refers to a clinical syndrome characterized by elevated levels of thyroid hormones in circulation, primarily manifesting as increased sympathetic nervous system excitability and metabolic hyperactivity. Thyrotoxicosis encompasses two conditions: hyperthyroidism and non-hyperthyroid thyrotoxicosis. Hyperthyroidism refers to thyrotoxicosis caused by excessive production of thyroid hormones by the thyroid gland. In contrast, non-hyperthyroid thyrotoxicosis arises when the thyroid hormone synthesis function is not increased but instead results from thyroid tissue destruction, releasing stored thyroid hormones into circulation, or excessive intake of exogenous thyroid hormones. Based on the severity of hyperthyroidism, the condition can be categorized into overt hyperthyroidism and subclinical hyperthyroidism.

Table 1 Common causes of thyrotoxicosis
Graves' disease (GD) is a multisystem autoimmune disorder characterized by the presence of thyrotropin receptor antibodies (TRAb) and represents the primary cause of hyperthyroidism. Women with Graves' hyperthyroidism are four times more likely to be affected than men, while the condition is relatively rare in children. The incidence tends to stabilize after the age of 30, with a mean age of onset of 47 years. This section focuses on the discussion of Graves' disease.
Etiology and Pathogenesis
Graves' disease develops in the context of a specific genetic predisposition under the influence of environmental factors, leading to an autoimmune response targeting the thyroid-stimulating hormone receptor (TSHR). Its development involves gene polymorphisms, including those of HLA, CTLA-4, PTPN22, CD40, IL-2R, FCRL3, Tg, and TSHR, as well as processes like X-chromosome inactivation skewing. Environmental factors, such as infections, iodine intake, smoking, stress, medications, and environmental toxins, also play a role.
TSHR is the primary autoantigen in Graves' disease. Lymphocytes infiltrating the thyroid gland produce thyroid autoantibodies. Following disruption of immune tolerance, the TSHRα subunit activates B lymphocytes, transforming them into plasma cells capable of secreting TSHR antibodies, including TRAb.
TRAb includes three subtypes:
- Thyroid-stimulating antibodies (TSAb)
- Thyroid-blocking antibodies (TBAb)
- Neutral antibodies
TSAb is the pathogenic antibody responsible for hyperthyroidism in Graves' disease. TSAb competes with thyroid-stimulating hormone (TSH) for binding to the α subunit of the TSH receptor (TSHR), activating the adenylate cyclase signaling pathway. This activation leads to the proliferation of thyroid follicular epithelial cells and excessive production of thyroid hormones. The stimulation of TSAb on TSHR is not subject to the negative feedback regulation of the hypothalamic-pituitary-thyroid axis, leading to hyperthyroidism.
TBAb blocks the binding of TSH to TSHR, reducing thyroid hormone synthesis. In patients with Graves' disease, the two antibodies can transform into one another, and the dominant antibody determines thyroid function. Neutral antibodies do not affect thyroid function.
Pathology
The thyroid gland exhibits varying degrees of diffuse enlargement. Thyroid follicular epithelial cells show increased proliferation, appearing as tall columnar or cuboidal cells, with the colloid in the follicular lumen reduced or absent. Infiltrating T cells, B cells, and plasma cells may be observed between the follicles.
In patients with Graves' ophthalmopathy, T lymphocytes infiltrate the retro-orbital tissue, adipocyte numbers increase, and fibroblasts secrete large amounts of mucopolysaccharides and glycosaminoglycans (e.g., hyaluronic acid). These changes lead to extraocular muscle swelling and proptosis. TSHR and insulin-like growth factor-1 (IGF-1) receptors exist on the surface of fibroblasts and adipocytes in the retro-orbital tissue. Activation of both receptors involves a shared signaling pathway.
Pre-tibial myxedema is characterized by extracellular matrix accumulation of hyaluronic acid, mucopolysaccharides, and lymphocyte infiltration. Swelling, fibroblast proliferation, and muscle fiber destruction may be observed in muscle tissue.
Clinical Manifestations
Symptoms of Thyrotoxicosis
Hypermetabolic Syndrome
Elevated thyroid hormones stimulate the sympathetic nervous system, enhance metabolism, and increase sensitivity to catecholamines. Catabolism of proteins, glucose, and fats exceeds anabolism, leading to increased heat production. Patients commonly experience diaphoresis, heat intolerance, increased appetite, weight loss, fatigue, abnormal glucose tolerance, or worsening of pre-existing diabetes.
Psychiatric and Neurological Symptoms
Common manifestations include irritability, emotional lability, insomnia, hyperactivity, and difficulty concentrating. Severe cases may present with delusions. Fine tremors may be observed when the patient extends their tongue or holds their arms outstretched. Reflexes are brisk, with shortened recovery phases for deep tendon reflexes.
Cardiovascular System
Enhanced metabolic activity and heat dissipation elevate cardiovascular workload, resulting in increased circulation. Symptoms include persistent palpitations, even at rest. On auscultation, findings may include tachycardia, accentuated first heart sound, arrhythmias, and systolic murmurs. Common arrhythmias include sinus tachycardia and atrial premature beats, followed by paroxysmal or persistent atrial fibrillation, with occasional ventricular or junctional premature beats, and rarely, atrioventricular conduction block. Systolic blood pressure elevation, diastolic pressure reduction, and an increased pulse pressure may lead to peripheral vascular signs such as capillary pulsation and collapsing pulse. Thyrotoxic heart failure, characterized as high-output cardiac failure, may arise or worsen following arrhythmias. Heart failure often improves or resolves with the resolution of hyperthyroidism.
Thyrotoxic heart disease refers to cardiac conditions caused by prolonged untreated hyperthyroidism. Manifestations may include:
- Severe arrhythmias such as persistent or paroxysmal atrial fibrillation, atrial flutter, frequent ventricular premature beats, or second- and third-degree atrioventricular block;
- Heart failure;
- Cardiac enlargement;
- Angina pectoris or myocardial infarction. Other cardiac diseases, such as hypertensive cardiomyopathy, coronary artery disease, and rheumatic heart disease, must be excluded.
Digestive System
Symptoms include frequent hunger, polyphagia, and weight loss. Accelerated gastrointestinal motility often results in increased bowel movements. Liver dysfunction may occur, such as elevated transaminase and alkaline phosphatase levels, hypoalbuminemia, and occasionally jaundice.
Musculoskeletal System
Thyrotoxic myopathy can be categorized into acute and chronic forms. Acute myopathy presents over a few weeks with swallowing difficulty and respiratory muscle paralysis. Chronic myopathy predominantly affects proximal muscle groups, such as the shoulders and hips, causing progressive muscle weakness with muscle atrophy, and difficulty climbing stairs, squatting, or combing hair. Thyrotoxic periodic paralysis (TPP) manifests as limb paralysis, primarily in the lower extremities, impairing standing or walking. It frequently co-occurs with hypokalemia due to intracellular shifts of potassium. TPP is self-limiting and resolves upon potassium supplementation. It is most commonly seen in young men of Asian and Latin American descent. Graves' disease (GD) may also cause thymic enlargement, which resolves after controlling hyperthyroidism. GD can coexist with myasthenia gravis.
Bone turnover increases in thyrotoxicosis, resulting in elevated urinary excretion of calcium, phosphorus, and collagen degradation products. Reduced serum 25-hydroxyvitamin D3 levels may lead to decreased bone mass and osteoporosis.
Hematopoietic System
Total leukocyte and neutrophil counts are reduced, while lymphocyte proportions are elevated. Erythrocyte numbers are often increased, with normal platelet counts and coagulation mechanisms. Occasional cases are associated with idiopathic thrombocytopenic purpura or pernicious anemia.
Reproductive System
Women commonly experience menstrual irregularities, prolonged cycles, amenorrhea, or infertility. Men may present with erectile dysfunction, and rarely, gynecomastia may be observed.
Skin and Hair
Vasodilation and excessive sweating cause the skin to feel warm and moist. Symptoms may include depigmentation, vitiligo, and hair loss.
Ophthalmic Symptoms
Eye symptoms are related to increased sympathetic activity caused by thyrotoxicosis. These include mild exophthalmos, mild eyelid retraction, widened palpebral fissures, a staring appearance, and decreased blinking. Eyelid lag refers to the upper eyelid failing to follow the eyeball promptly when looking downward. Lag of the globe occurs when the upward movement of the eye lags behind the upper eyelid. Impaired or insufficient convergence during near-focus (Mobius sign) is another observation.
Thyroid Storm (Thyroid Crisis)
Thyroid storm is a critical condition with high mortality, characterized by an acute exacerbation of thyrotoxic symptoms. It often occurs in patients with poorly managed or untreated severe hyperthyroidism. Common triggers include infection, trauma, emotional stress, radioactive iodine therapy, thyroid surgery, or postpartum. Clinical features include hyperpyrexia, profuse sweating, tachycardia with heart rates typically exceeding 140 beats per minute, irritability, anxiety, delirium, nausea, emesis, diarrhea, and severe complications such as heart failure, shock, and coma. Diagnosis is based on the comprehensive assessment of clinical manifestations. Early recognition of pre-crisis symptoms allows urgent management as thyroid storm.
Apathetic Hyperthyroidism
This form is more common in elderly patients and has an insidious onset. Symptoms may include congestive heart failure with arrhythmias, unexplained weight loss, and subtle hypermetabolic signs. It is often misdiagnosed as malignancy, coronary artery disease, or other illnesses.
Characteristic Features of Graves' Disease
Thyroid Enlargement
The thyroid gland shows diffuse enlargement with a soft or firm consistency and no tenderness. In typical cases, a fine thrill may be palpable at the upper and lower poles of the thyroid gland, and vascular bruits may be audible on auscultation.
Graves' Ophthalmopathy (GO)
Graves' ophthalmopathy (GO), also referred to as thyroid-associated ophthalmopathy (TAO) or thyroid eye disease (TED), occurs in 25%–50% of patients with Graves' disease (GD). Among these, 10%–20% have unilateral involvement. Hyperthyroidism and GO may occur simultaneously or sequentially. Approximately 5% of GO patients exhibit normal thyroid function, a condition termed euthyroid Graves' ophthalmopathy (EGO). While GO is more common in women, its clinical presentation is more severe in men.
Symptoms include a foreign body sensation in the eyes, discomfort or pain, photophobia, tearing, diplopia, strabismus, and impaired vision. Clinical findings may include unilateral or bilateral eyelid retraction, proptosis, eyelid erythema and swelling, conjunctival hyperemia and edema, caruncular swelling, restricted eye movements, or in severe cases, fixation of the eyeball. Incomplete eyelid closure and corneal exposure can result in exposure keratopathy. Posterior orbital compression of the optic nerve can lead to dysthyroid optic neuropathy (DON) in GO, which, along with severe ocular complications, may result in vision loss.
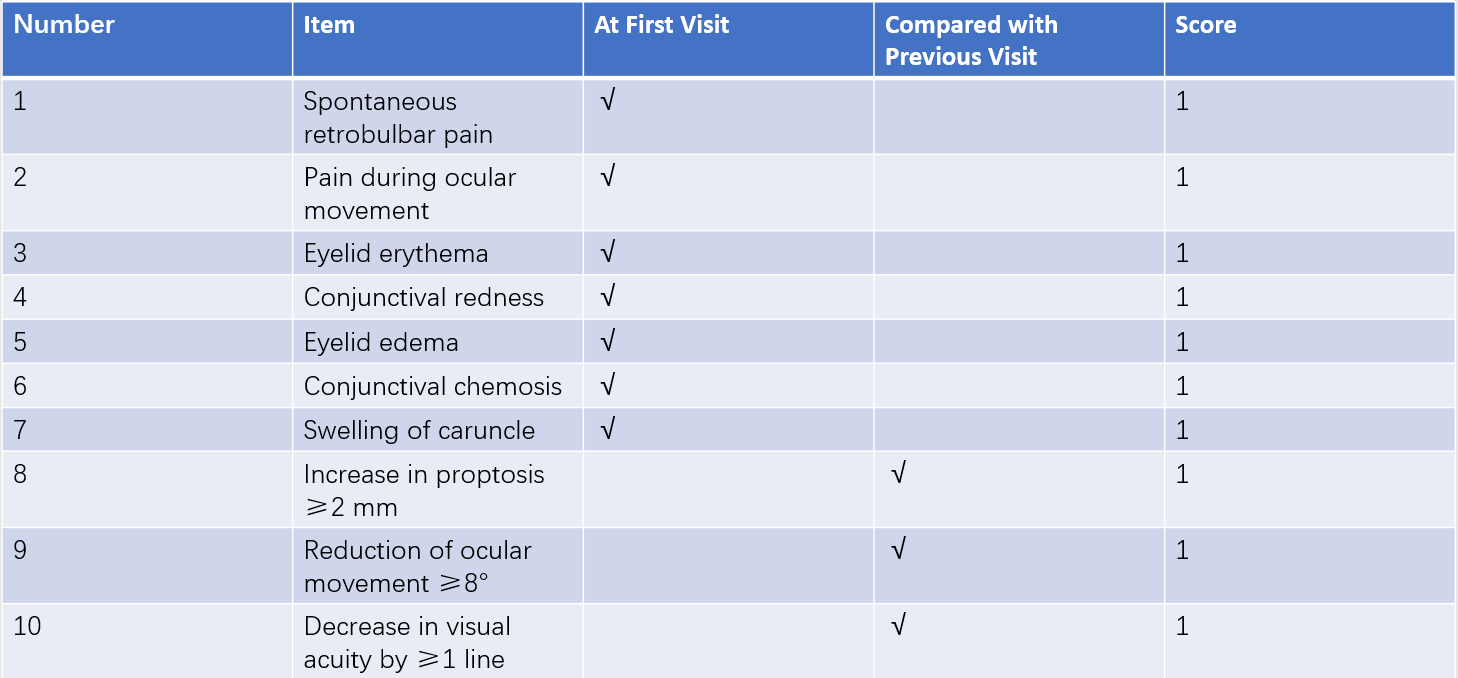
Table 2 Clinical activity score (CAS) for GO (EUGOGO, 2021)
Notes: CAS is based on criteria provided by EUGOGO (European Group on Graves’ Orbitopathy).

Table 3 Severity classification of GO (American Thyroid Association)
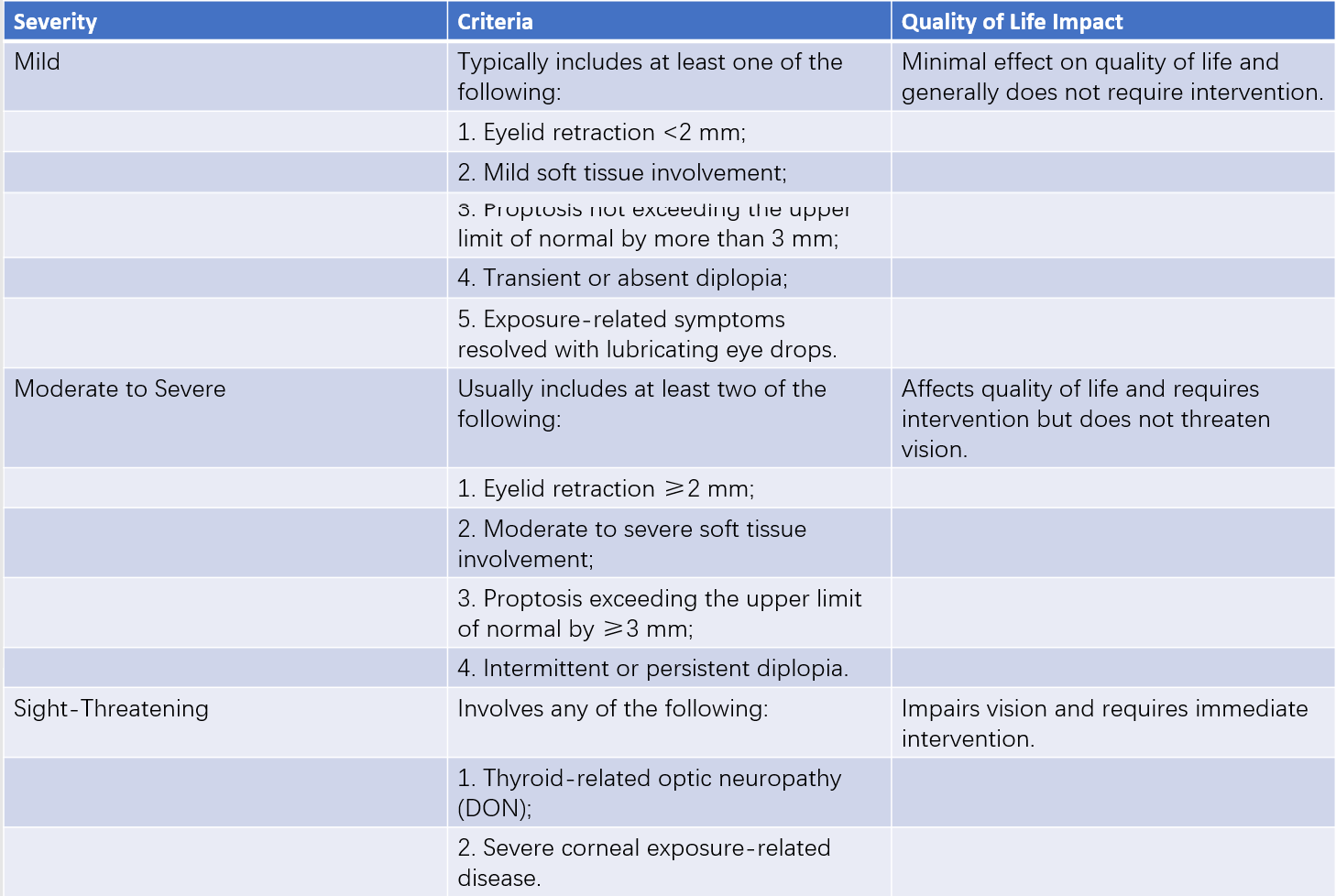
Table 4 Severity classification of GO (EUGOGO, 2021)
Evaluation of clinical activity stages and severity classification is essential in all GO patients to guide the management of both hyperthyroidism and GO. Clinical activity staging uses the clinical activity score (CAS). A CAS of ≥3 points (in the initial 7-point evaluation) or ≥4 points (in subsequent 10-point evaluations) indicates active GO. GO is considered inactive if the scores fall below these thresholds. There are two main systems to classify the severity of GO.
Graves' Dermopathy
Pretibial myxedema (PTM) is observed in a minority of GD patients and is more common in Caucasians. It often occurs in the lower one-third of the anterior tibia but may also be found around the ankles, on the dorsum of the feet, shoulders, dorsum of the hands, surgical scars, or, rarely, on the face. The condition typically presents symmetrically.
Early changes include localized skin thickening and roughening, with reddish-brown plaques or nodules of varying sizes that enlarge and gradually merge. In advanced stages, the skin takes on an orange-peel or bark-like appearance, often covered with gray or black wart-like lesions. The lower legs may become markedly enlarged, resembling elephantiasis. Acropachy, a rare manifestation, involves the thickening and swelling of the distal fingers and toes in a club-like manner, with overlying skin thickening.
Laboratory and Other Examinations
Thyroid-Stimulating Hormone (TSH)
Serum TSH is the most sensitive marker for assessing thyroid function. The use of highly sensitive TSH assays enables the diagnosis of subclinical hyperthyroidism, making TSH the first-line screening test for hyperthyroidism.
Serum Total Thyroid Hormones
All T4 in the blood is produced by the thyroid gland, with daily production around 80–100 μg, while only 20% of T3 is produced directly by the thyroid gland, with the remaining 80% converted from T4 in peripheral tissues via deiodinase enzymes. In the bloodstream, 99.96% of T4 and more than 99.5% of T3 are protein-bound to form bound thyroid hormones. Of these, 80%–90% bind to thyroxine-binding globulin (TBG), followed by transthyretin and albumin. Total thyroid hormones measure both bound and free thyroid hormones. Changes in serum TBG concentration or the protein-hormone binding capacity can influence total thyroid hormone levels. For example, pregnancy, estrogen use, acute viral hepatitis, or genetic factors may increase TBG levels, causing elevated TT4 and TT3 levels; meanwhile, androgens, glucocorticoids, or certain genetic factors may reduce TBG levels, decreasing TT4 and TT3 levels.
Serum Free Thyroid Hormones
These include free thyroxine (FT4) and free triiodothyronine (FT3). Although free thyroid hormones represent only a minute portion of circulating thyroid hormones, they are the biologically active forms and serve as key indicators for diagnosing clinical hyperthyroidism. In most hyperthyroid cases, both FT3 and FT4 are elevated, with FT3 potentially rising earlier than FT4. When only FT3 is elevated but FT4 is normal, the condition is referred to as T3 toxicosis, which is often seen in the early stages of hyperthyroidism, during hyperthyroidism treatment, or in relapse after remission. Conversely, when only FT4 is elevated and FT3 is normal, the condition is referred to as T4 toxicosis.
Thyroid-Stimulating Hormone Receptor Antibodies (TRAb)
In untreated Graves' disease (GD) patients, TRAb tests yield a positive rate of 80%–100%. TRAb serves as an indicator for distinguishing hyperthyroidism etiology and diagnosing GD, and provides guidance in evaluating disease activity, deciding on treatment discontinuation, and assessing post-treatment recurrence risk. Currently, clinical TRAb tests only detect the presence of autoantibodies targeting the TSH receptor, without distinguishing the functional type of antibody, i.e., stimulating antibodies (TSAb) or blocking antibodies (TBAb). For simplicity, TRAb is often considered equivalent to TSAb in hyperthyroid states. Differentiation between TSAb and TBAb can be achieved by adding serum to cell lines transfected with human TSH receptors and measuring cAMP levels in the culture medium.
TRH Stimulation Test
Sensitive TSH assays have largely supplanted the TRH stimulation test for diagnosing hyperthyroidism. In thyroid-origin hyperthyroidism, elevated T3 and T4 levels suppress TSH secretion; lack of a TSH response to TRH stimulation supports a diagnosis of thyroidal hyperthyroidism.
Radioactive Iodine Uptake (RAIU)
Although RAIU has traditionally been used to diagnose hyperthyroidism, it has largely been replaced by sensitive TSH testing. Normal RAIU values vary by region but typically range from 5%–25% at 3 hours and 20%–45% at 24 hours, with the peak uptake occurring at 24 hours. RAIU remains valuable for distinguishing the etiology of thyrotoxicosis: in hyperthyroidism, serum thyroid hormone levels are elevated alongside increased or normal RAIU, with an advanced uptake peak. In contrast, destruction-induced thyrotoxicosis presents with elevated serum thyroid hormone levels but reduced RAIU, leading to a dissociation phenomenon. RAIU is also used to calculate required doses of radioactive iodine (131I) for treating hyperthyroidism. RAIU testing is contraindicated during pregnancy and lactation.
Thyroid Radionuclide Scanning
This method is useful for diagnosing toxic thyroid nodules and autonomously functioning thyroid adenomas. Functioning nodules or tumor areas show increased radionuclide uptake (hot nodules), while non-nodular thyroid tissue exhibits low or no radionuclide uptake.
Thyroid Ultrasound
In GD, ultrasound typically reveals thyroid enlargement and significantly increased blood flow. Peak systolic blood flow velocity in the superior and inferior thyroid arteries is elevated. Ultrasound can assist in distinguishing thyrotoxicosis caused by thyroid destruction.
CT and MRI
Orbital CT and MRI are utilized to assess the extent of extraocular muscle involvement, aiding the diagnosis of Graves' ophthalmopathy (GO). These imaging modalities help evaluate clinical activity staging, severity grading, and distinguish GO from other causes of exophthalmos.
Diagnosis
Diagnosis encompasses both thyrotoxicosis and the identification of its underlying cause. Typical cases are usually straightforward, while atypical ones are prone to misdiagnosis or underdiagnosis.
Diagnosis of Thyrotoxicosis and Hyperthyroidism
Elevated serum FT3 or FT4 levels, combined with clinical symptoms, confirm thyrotoxicosis. Increased RAIU or heightened radionuclide uptake in static thyroid imaging supports the diagnosis of hyperthyroidism. Reduced TSH with elevated FT3 or FT4 confirms clinical hyperthyroidism; reduced TSH with normal T3 and T4 confirms subclinical hyperthyroidism.
Diagnosis of Graves' Disease (GD)
Criteria for GD diagnosis include:
- Confirmed hyperthyroidism;
- Diffuse thyroid enlargement (some cases may lack palpable goiter);
- Presence of Graves' ophthalmopathy;
- Pretibial myxedema or digital clubbing;
- Elevated TRAb levels.
Criteria 1 and 2 are essential for diagnosis. GD can be diagnosed if one of criteria 3, 4, or 5 is also met, based on characteristic clinical features of Graves' disease and TRAb findings.
Differential Diagnosis
Distinguishing GD from Destructive Thyrotoxicosis
Destructive thyrotoxicosis (e.g., subacute thyroiditis, painless thyroiditis) presents with symptoms of hypermetabolism and elevated serum thyroid hormone levels. However, the hallmark is reduced thyroid radioactive iodine uptake (RAIU), creating a "dissociation phenomenon."
Distinguishing GD from Other Causes of Hyperthyroidism
In toxic multinodular goiter and thyroid autonomously functioning adenoma, thyroid ultrasound often reveals nodules or tumors. Thyroid radionuclide scanning in multinodular goiter shows uneven radionuclide distribution with patchy areas of hyperactivity and hypoactivity, while autonomously functioning adenoma appears as "hot" nodules with suppressed tracer uptake in surrounding thyroid tissue. Hashitoxicosis can be difficult to differentiate and may require thyroid biopsy or follow-up to observe disease progression. Hyperthyroidism associated with human chorionic gonadotropin (HCG) manifests with significantly elevated blood HCG levels and related conditions. Iodine-induced hyperthyroidism is characterized by a history of excessive iodine intake and reduced RAIU levels.
Distinguishing GD from Gestational Transient Thyrotoxicosis (GTT)
GTT occurs early in pregnancy due to high levels of HCG stimulating thyroid TSH receptors. The condition is mild, transient, and improves with declining HCG levels in the second or third trimester. It is more likely in cases of hyperemesis gravidarum or multiple gestations. GTT does not require treatment with antithyroid drugs (ATDs).
Distinguishing GD from Other Diseases
Conditions Mimicking Thyrotoxicosis Symptoms
Premenopausal women may experience emotional instability, irritability, insomnia, hot flashes, and perspiration, resembling thyrotoxicosis. Diabetes mellitus may present with increased appetite, hunger, and weight loss. Patients with tuberculosis, chronic infections, or malignancies may exhibit weight loss, low fever, and cachexia, while primary myopathies can cause severe muscle atrophy. Thyroid function testing is essential for differentiation. Conditions such as congestive heart failure and refractory atrial fibrillation that respond poorly to standard treatment should raise suspicion for hyperthyroidism.
Conditions Mimicking GD Signs
Simple goiter causes thyroid gland enlargement without hyperthyroid symptoms, and tests show normal blood TSH, T4, and T3 levels. RAIU may be elevated, but the uptake peak does not shift earlier. For patients with exophthalmos, other causes such as intracranial tumors, carotid-cavernous sinus fistula, orbital cellulitis, or orbital tumors should be considered. CT or MRI imaging can confirm the diagnosis.
Treatment
Currently, there is no definitive etiologic treatment for GD. Classic therapeutic options include antithyroid drugs (ATDs), radioactive iodine (131I), and surgery, each with its advantages and disadvantages. ATDs are the most commonly used treatment.
Antithyroid Drugs (ATDs)
ATDs are thioamide compounds that include imidazoles (e.g., methimazole [MMI] and carbimazole) and thiouracils (e.g., propylthiouracil [PTU] and methyloxime uracil). MMI and PTU are commonly used. Both MMI and PTU are primary treatments for GD. MMI is generally the first choice, except in the following situations where PTU may be preferred: early pregnancy, thyroid storm management, or when MMI is poorly tolerated and the patient refuses 131I or surgery. The mechanism of ATDs involves inhibition of thyroid peroxidase (TPO) activity, thereby blocking iodination and the coupling of iodotyrosines, reducing thyroid hormone synthesis. ATDs may also have immunosuppressive effects, lowering TRAb levels.
The cure rate of GD with ATDs is approximately 50%, with recurrence rates ranging from 40% to 60%. A minority of patients may require long-term low-dose maintenance therapy.
Indications
ATDs are suitable for the initial treatment of all hyperthyroid patients, preoperative preparation for surgery, and pre- or post-131I therapy. Patients with mild hyperthyroidism, small thyroid glands, or TRAb levels that are negative or mildly elevated are more likely to achieve remission with ATDs. Elderly patients with poor general health who cannot tolerate surgery, those with a limited life expectancy, patients with a history of recurrent hyperthyroidism following surgery or contraindications to 131I therapy, those requiring rapid control of thyroid function, and patients with moderate-to-severe active GO may be considered for ATD therapy as the first choice.
Dosing and Treatment Phases
ATD therapy is divided into three phases: the initial treatment phase, dose reduction phase, and maintenance phase. Drug dosage is adjusted based on the severity of the condition.
Initial Treatment Phase
Typical dosing for MMI is 10–30 mg/day, with a plasma half-life of 6 hours, given once daily or divided into three doses. PTU dosing is 100–300 mg/day, with a plasma half-life of 1.5 hours, administered in three divided doses. Thyroid function is reassessed every 4 weeks, and the dosage is reduced as T3 and T4 levels approach normal.
Dose Reduction Phase
Dosage reductions are made every 2–4 weeks. For MMI, the dose is decreased by 5–10 mg per adjustment, while PTU is reduced by 50–100 mg each time. Thyroid function is reassessed every 4 weeks, and once TSH levels normalize, the dosage is gradually reduced to the minimum maintenance dose.
Maintenance Phase
Maintenance doses are typically 5–10 mg/day for MMI and 50–100 mg/day (or less) for PTU. Thyroid function is monitored every 3–6 months. Unless adverse reactions occur, ATD therapy is generally not interrupted, and efficacy is periodically assessed. If hypothyroidism develops during treatment, the ATD dose should be reduced, or levothyroxine (L-T4) may be added as needed.
Treatment duration typically spans 18–24 months. Discontinuation of ATDs is considered when the following criteria are met: full treatment course, normalization of TRAb, and the ability to maintain normal TSH levels with minimal ATD doses.
Efficacy of ATD Treatment
Remission of hyperthyroidism refers to the normalization of TSH and thyroid hormone levels for at least one year after ATD discontinuation. Relapse of hyperthyroidism is defined as the recurrence of hyperthyroidism more than one year following remission. Factors contributing to relapse include male gender, smoking, elevated TRAb levels, significant thyroid enlargement, and increased blood flow within the thyroid gland. There are no reliable clinical markers for predicting relapse. Normalization of TRAb levels and prolonged low-dose ATD therapy might help reduce the risk of relapse. Patients experiencing relapse may consider retreatment with ATD, radioactive iodine (131I), or surgery.
Side Effects of ATDs
Rashes occur in approximately 5% of patients. Mild rashes may improve with antihistamines, while severe rashes require discontinuation of the drug and switching to another ATD, 131I treatment, or surgery.
Leukopenia is observed in around 5% of patients, while agranulocytosis has an incidence of about 0.37%. These conditions often occur within 2–3 months of treatment but can develop at any time. Discontinuation of the drug should be considered if the peripheral white blood cell count drops below 3×109/L or neutrophils fall below 1.5×109/L. Agranulocytosis, defined as a neutrophil count below 0.5×10⁹/L, requires immediate medical intervention. Due to cross-reactivity between drugs, switching to another ATD is not recommended in these cases. Differentiation between leukopenia caused by hyperthyroidism or ATD use necessitates routine monitoring of total white blood cell and neutrophil counts before and during treatment. Clinical symptoms such as fever and sore throat should also be assessed.
Mild hepatic injury is common, while severe cases occur at an incidence of 0.1%–0.2%. Because hyperthyroidism itself may cause liver dysfunction, liver function monitoring is recommended before and during ATD therapy. PTU primarily causes hepatocellular damage, unrelated to dose, and can result in acute liver failure in severe cases. MMI is more likely to induce cholestasis, and its effects are dose-dependent.
Anti-neutrophil cytoplasmic antibody (ANCA)-associated vasculitis is observed mainly in women receiving long-term PTU therapy. Symptoms include fever, joint pain, and muscle soreness, while severe cases may involve hemoptysis, dyspnea, and hematuria.
Radioactive Iodine (131I) Therapy
During hyperthyroidism, the thyroid's iodine uptake capacity increases, allowing 131I to be absorbed and release β-rays that destroy thyroid follicular cells, thereby reducing thyroid hormone production and alleviating hyperthyroidism.
Indications and Contraindications
131I therapy is particularly suitable for the following patients:
- Failure of ATD therapy or the emergence of severe adverse reactions.
- Contraindications or high risks associated with surgery.
- History of neck surgery or external radiation.
- Patients with liver dysfunction, leukopenia, thrombocytopenia, periodic paralysis, atrial fibrillation, or diabetes.
- Patients with a long disease course.
- Elderly patients, especially those with cardiovascular comorbidities.
Women of childbearing age are advised to consider pregnancy only six months after treatment. For patients with mild active Graves' ophthalmopathy (GO) or moderate-to-severe non-active GO, the addition of corticosteroids after 131I therapy may be beneficial. Contraindications include pregnancy, lactation, and confirmed or suspected thyroid cancer. Patients with moderate-to-severe active GO should use 131I therapy with caution.
Dosage
Two methods are used for determining 131I dosage:
- Calculated Dose: Based on estimated thyroid gland volume and RAIU. Typically, a dose of 2.59–5.55 MBq (70–150 μCi) per gram of thyroid tissue is administered.
- Fixed Dose: A fixed dose is administered based on thyroid volume. Severe cases may benefit from pre-treatment with ATDs to alleviate symptoms, followed by 131I ingestion after ATD discontinuation. For severe GD cases, ATDs may be used for short periods after 131I therapy.
Efficacy
The cure rate for hyperthyroidism with 131I therapy exceeds 85%. Symptoms tend to improve, and thyroid size decreases within 2–4 weeks after treatment. Thyroid function typically normalizes within 6–12 weeks. If symptoms persist or treatment is ineffective after three months, another course of 131I therapy may be administered. Both the normalization of thyroid function and the onset of hypothyroidism are considered successful outcomes. Hormone replacement therapy with thyroid hormones is required if hypothyroidism develops.
Complications
Radiation thyroiditis typically occurs 7–10 days after treatment.
Thyroid storm mainly arises in patients with uncontrolled severe hyperthyroidism.
The risk of exacerbating active GO can be mitigated with corticosteroid therapy following 131I treatment.
Surgical Treatment
Indications
Indications include:
- Moderate-to-severe hyperthyroidism unresponsive to standard ATD therapy or in patients unwilling to continue long-term medication.
- Significant thyroid enlargement causing compressive symptoms.
- Hyperthyroidism with retrosternal goiter.
- Severe adverse reactions to ATDs or reluctance/inappropriateness for 131I therapy or its ineffectiveness.
- Co-occurrence of thyroid malignancy or primary hyperparathyroidism.
- Presence of moderate-to-severe GO unresponsive to medical treatment.
- Patient preference for surgery to shorten treatment duration or to achieve rapid symptom relief.
Contraindications
Contraindications include:
- Severe comorbidities or poor general health that precludes surgery.
- Early or late pregnancy.
Preoperative Preparation
Patients should undergo adequate ATD and β-blocker treatment prior to surgery until symptoms are well-controlled, heart rate falls below 80 beats per minute, and T3 and T4 levels normalize. Starting two weeks pre-surgery, Lugol's iodine solution (3–5 drops per dose, once to three times daily) may be administered to reduce intraoperative bleeding.
Surgical Procedure and Complications
The preferred procedure is total or near-total thyroidectomy. Surgical complications may include wound bleeding, respiratory obstruction, infection, recurrent laryngeal nerve injury, temporary or permanent hypoparathyroidism, and hypothyroidism. Severe complications may lead to a thyroid storm.
Other Treatments
General Management
Adequate rest and sufficient intake of calories and nutrients, including carbohydrates, proteins, and B vitamins, are recommended. Sedatives may be used for patients with mental stress, anxiety, or insomnia. Dietary iodine intake should be moderately restricted, and iodine-containing medications or contrast agents should be avoided. Compound iodine solution should be used only in preoperative preparation or during thyroid storm.
Beta Blockers
Beta blockers antagonize beta-adrenergic receptors and alleviate the effects of catecholamines. In high doses, propranolol can inhibit the peripheral conversion of T4 to T3. These drugs are used as adjunctive therapy in the initial stages of hyperthyroidism to control clinical symptoms. Non-selective beta blockers should not be used in patients with bronchial asthma or asthmatic bronchitis.
Treatment of Thyroid Storm
Prompt and aggressive management is required for suspected or confirmed thyroid storm.
- Inhibition of Thyroid Hormone Synthesis: This is the first step in treatment. High doses of PTU, which can also inhibit the peripheral conversion of T4 to T3, are the preferred option for thyroid storm. The recommended PTU dose is 600 mg daily, divided into multiple oral doses or administered via a gastric tube, with a maximum daily dose of 1,200 mg. If PTU is unavailable or contraindicated due to allergy, MMI may be used at a daily dose of 60 mg, with a maximum of 120 mg. Dosages are adjusted based on the patient’s condition, transitioning to standard therapeutic doses after symptom relief.
- Inhibition of Thyroid Hormone Release: One to two hours after PTU administration, inorganic iodide (Lugol's solution) can be added, with a dosage of 4–8 drops every 6–8 hours. The dosage is gradually reduced depending on clinical improvement, and treatment is typically discontinued after 3–7 days. High-dose iodine also inhibits thyroid hormone synthesis.
- Inhibition of T4-to-T3 Conversion: High-dose PTU, iodide, propranolol, and corticosteroids can reduce T4-to-T3 conversion. These agents may be combined, if no contraindications are present, to enhance therapeutic efficacy. Propranolol is administered orally at 60–80 mg every 4–6 hours. Hydrocortisone (50–100 mg) or dexamethasone (2 mg) can be given via intravenous infusion every 6–8 hours, with gradual tapering and discontinuation as the patient improves.
- Reduction of Circulating Thyroid Hormone Levels: If standard treatments fail to achieve satisfactory results, procedures such as plasma exchange or hemodialysis may be considered.
- Supportive Therapy: Monitoring of cardiac, renal, and cerebral function is essential. Electrolyte imbalances and acid-base disorders are corrected, while adequate glucose, calories, and multiple vitamins are supplied.
- Symptomatic Management: Oxygen therapy and infection prevention and management are provided. High fever can be treated with physical cooling methods, although salicylate medications, such as aspirin, should be avoided.
Treatment of Graves’ Ophthalmopathy (GO)
General Measures
For all GO patients, risk factors, such as smoking and hypercholesterolemia, should be managed. Thyroid function is maintained at normal levels by choosing appropriate hyperthyroidism treatments. Local symptomatic treatments include the use of tinted glasses, artificial tears, and eye shields for corneal protection. The patient is advised to sleep with the head elevated.
Specific Treatments for GO
Treatment options for GO include medications, orbital radiotherapy, and surgery, with decisions based on disease activity, severity, safety, cost, and patient preferences.
Mild GO
During the active phase, patients may be monitored if risk factors are addressed, or they may receive selenium supplementation for 6 months. In the stable phase, observation or rehabilitative eye surgery may be considered if necessary.
Moderate-to-Severe GO
Active-phase treatment options include corticosteroids, targeted immunosuppressants, orbital radiotherapy, and orbital decompression surgery, with earlier interventions yielding better outcomes. In the stable phase, rehabilitative eye surgery may be performed if needed.
Corticosteroids
Intravenous therapy is more effective than oral or local treatments. Pulse therapy using methylprednisolone (500 mg diluted in saline, administered weekly for 6 weeks, followed by 250 mg weekly for an additional 6 weeks) is commonly used, resulting in a total treatment duration of 12 weeks. For more severe active GO, a higher-dose pulse protocol is used (0.75 g per dose weekly for 6 weeks, followed by 0.5 g weekly for 6 weeks, with a cumulative dose of 7.5 g). The total corticosteroid dose should not exceed 6.0–8.0 g. Monitoring and managing medication side effects is essential during treatment.
Orbital Radiotherapy
This can be used alone or in combination with corticosteroids, improving globe motility, restrictive strabismus, and diplopia. Commonly used regimens include a total dose of 20 Gy, delivered as 2 Gy per session for 10 consecutive days (completed within 2 weeks), or as 1 Gy per session weekly for 20 weeks. Orbital radiotherapy is contraindicated in pregnant women and patients with diabetic retinopathy.
Orbital Decompression Surgery
This includes fat decompression and bony wall decompression, which aim to increase orbital volume and relieve optic nerve compression.
Other Medications
Monoclonal antibodies such as teprotumumab (anti-IGF-1 receptor antibody), tocilizumab (anti-IL-6 receptor antibody), and rituximab (anti-CD20 monoclonal antibody) are effective for treating moderate-to-severe active GO. Conventional immunosuppressants, including cyclosporine, methotrexate, and azathioprine, may also be used in combination with corticosteroids or orbital radiotherapy.
Vision-threatening GO
Severe vision loss caused by corneal exposure or thyroid-associated optic neuropathy (DON) requires urgent treatment. For DON, high-dose intravenous methylprednisolone (500–1,000 mg per dose, administered daily for 3 consecutive days or every other day for 1 week) is recommended for 2 weeks. Further treatment with sequential intravenous methylprednisolone or emergency orbital decompression surgery is determined by response to initial therapy.
Management of Hyperthyroidism During Pregnancy
Hyperthyroidism increases the risk of complications for both pregnant women and the fetus, including miscarriage, preterm delivery, pregnancy-induced hypertension, low birth weight, and intrauterine growth restriction. Elevated HCG levels during early pregnancy may exacerbate hyperthyroidism. Pregnancy is preferably planned when thyroid function—ideally TRAb levels—is normal or at low levels to reduce the risk of fetal or neonatal hyperthyroidism (maternal TRAb can cross the placenta).
PTU is preferred during pregnancy because it binds to proteins and has limited placental transfer. The smallest effective dose is used to maintain maternal FT4 levels at the high-normal or slightly elevated range, reducing the risk of fetal hypothyroidism or goiter due to overtreatment.
For lactating mothers requiring continued ATD therapy, both PTU and MMI are considered safe. Doses of MMI below 20 mg/day or PTU below 300 mg/day are generally safe, and the lowest effective dose is recommended.