Diabetes mellitus (DM) is a group of metabolic diseases characterized by chronic hyperglycemia caused by various etiologies. It results from defects in insulin secretion and/or action. Long-term disturbances in carbohydrate, fat, and protein metabolism can lead to multi-system damage, causing chronic progressive pathological changes, functional impairment, and failure of organs and tissues such as the eyes, kidneys, nerves, heart, and blood vessels. In severe cases or under stress, acute metabolic disorders such as diabetic ketoacidosis (DKA) and hyperosmolar hyperglycemic syndrome may occur.
Classification
The classification of diabetes mellitus is based on understanding its etiology, pathophysiology, and clinical manifestations. As knowledge of the disease has evolved, the classification system has become progressively refined, although it remains incomplete. Discrepancies and debates still exist between classifications recommended by different academic organizations, and the current classification system is expected to undergo further modifications, refinements, and updates in the future.
The following is the updated classification published by the World Health Organization (WHO) Diabetes Expert Committee in 2019:
Type 1 Diabetes Mellitus (T1DM)
This type is primarily characterized by immune-mediated destruction of pancreatic β-cells, often resulting in absolute insulin deficiency.
Type 2 Diabetes Mellitus (T2DM)
This type involves a progressive spectrum ranging from predominant insulin resistance with inadequate compensatory insulin secretion to predominant defects in insulin secretion accompanied by insulin resistance.
Hybrid Forms of Diabetes
Slowly Progressive Immune-Mediated Diabetes in Adults
This condition is similar to slowly progressive T1DM in adults but differs in that it is associated with features of metabolic syndrome, has greater preservation of pancreatic β-cell function, expresses a single autoantibody (particularly GAD65 antibody), and involves TCF7L2 gene polymorphisms.
Ketosis-Prone Type 2 Diabetes
A non-immune-mediated form of diabetes with a predisposition to ketosis, it is characterized by severe insulin deficiency during episodes of ketosis, followed by remission without the need for insulin therapy.
Other Specific Types of Diabetes
This category includes hyperglycemic states with relatively well-defined etiologies at different levels, ranging from environmental and genetic factors to their interactions.
Monogenic Diabetes
Monogenic Defects in Pancreatic β-Cell Function:
- Maturity-onset diabetes of the young (MODY)
- Mitochondrial gene mutation-related diabetes
- Neonatal diabetes
- Other forms
Monogenic Defects in Insulin Action:
- Insulin receptor gene mutations (e.g., Type A insulin resistance, Donohue syndrome, Rabson-Mendenhall syndrome)
- Familial partial lipodystrophy
- Congenital generalized lipodystrophy
- Other forms
Exocrine Pancreatic Diseases
These include diabetes secondary to conditions such as pancreatitis, trauma/surgical resection of the pancreas, pancreatic tumors, cystic fibrosis, hemochromatosis, and fibrocalcific pancreatic disease.
Endocrine Disorders
These include diabetes arising from conditions such as Cushing’s syndrome, acromegaly, pheochromocytoma, glucagonoma, hyperthyroidism, and somatostatinoma.
Diabetes Induced by Medications or Chemicals
This includes hyperglycemia caused by glucocorticoids, thyroid hormones, thiazide diuretics, α-adrenergic agonists, β-adrenergic agonists, phenytoin, pentamidine, niacin, rat poison, interferon-α, immune checkpoint inhibitors, and others.
Infections
These include congenital rubella, cytomegalovirus infection, and other infections.
Uncommon Immune-Mediated Diabetes
This includes stiff-person syndrome and insulin receptor autoantibodies.
Other Genetic Disorders Associated with Diabetes
These include disorders such as trisomy 21 (Down syndrome), Friedreich’s ataxia, Huntington’s disease, Klinefelter syndrome, Laurence-Moon-Biedl syndrome, myotonic dystrophy, porphyria, Prader-Willi syndrome, Turner syndrome, and others.
Unclassified Diabetes
Newly diagnosed cases of diabetes that cannot be definitively classified are initially categorized within this group.
Hyperglycemia First Detected During Pregnancy
This condition refers to hyperglycemic states diagnosed during pregnancy and is further classified into two categories:
Diabetes Mellitus in Pregnancy
This refers to diabetes first diagnosed during pregnancy, with diagnostic criteria identical to those outside of pregnancy.
Gestational Diabetes Mellitus (GDM)
This refers to abnormal glucose metabolism during pregnancy, diagnosed using criteria that are less stringent than those for diabetes in nonpregnant individuals.
Hyperglycemia first detected during pregnancy excludes women with preexisting or previously diagnosed diabetes, which is referred to as pregestational diabetes mellitus complicating pregnancy.
Among diabetes patients, T2DM accounts for the majority of cases, representing 90–95% of all diagnoses.
Etiology, Pathogenesis, and Natural History
The etiology and pathogenesis of diabetes mellitus are extremely complex and remain incompletely understood. Different types of diabetes have distinct etiologies, and there is heterogeneity even within the same type. In general, both genetic predisposition and environmental factors play roles in its development.
Insulin is synthesized and secreted by pancreatic β-cells, then reaches target cells in various tissues and organs through the bloodstream. It binds to specific receptors at the cell surface and elicits intracellular metabolic effects. Abnormalities in any part of this process may lead to diabetes mellitus.
The natural course of diabetes, regardless of its etiology, typically progresses through several stages. Patients may already have diabetes-related pathophysiological changes (such as the presence of autoantibodies, insulin resistance, or pancreatic β-cell dysfunction) for an extended period while maintaining normal glucose tolerance. With disease progression, impaired glucose regulation (IGR) may develop, which includes impaired fasting glucose (IFG) and/or impaired glucose tolerance (IGT). IGR represents an intermediate metabolic state between normal glucose homeostasis and diabetes-related hyperglycemia and is often referred to as "prediabetes." Eventually, the condition progresses to overt diabetes.
Type 1 Diabetes Mellitus (T1DM)
Most cases of T1DM are immune-mediated, involving the contribution of both genetic and environmental factors. Certain external factors (e.g., viral infections, chemical toxins, dietary factors) may act on genetically predisposed individuals, activating a series of T lymphocyte-mediated autoimmune responses that selectively destroy pancreatic β-cells and impair their function. This results in progressively reduced insulin secretion, eventually leading to diabetes.
Some cases of T1DM have unknown causes, with pancreatic β-cell destruction unrelated to human leukocyte antigen (HLA) or specific genetic types and lacking evidence of autoimmune responses. With the increasing prevalence of overweight and obesity in children and adolescents, some cases of T1DM also exhibit insulin resistance. Insulin resistance plays a role in the development and/or accelerated progression of T1DM.
Genetic Factors
The concordance rate for T1DM in monozygotic twins reaches 30–40%, indicating that genetic factors play an important role in its development. The genetic susceptibility to T1DM involves numerous genes, including HLA and non-HLA genes, though they remain incompletely identified. It is known that HLA, located on the short arm of chromosome 6, serves as a major susceptibility locus, contributing nearly 50% of genetic susceptibility, while others are considered minor susceptibility genes.
HLA class I and class II molecules respectively mediate the immune tolerance and immune damage of CD8+ and CD4+ T lymphocytes, thereby playing a role in T1DM pathogenesis. Specific HLA genotypes are closely associated with autoimmune responses targeting pancreatic β-cells, with HLA-DR and HLA-DQ being the most relevant loci in the HLA region. Susceptibility genes predispose individuals to develop autoantibodies and insulitis (inflammation of the islets), though these genetic factors alone are insufficient to cause overt diabetes.
Non-HLA genes may also contribute to T1DM susceptibility. Recent studies have identified numerous genes involved in regulating pancreatic β-cell apoptosis and insulin secretion, which play roles in the progression from insulitis to diabetes. Additionally, epigenetic mechanisms affecting gene expression and function may also be important in T1DM development. T1DM exhibits significant heterogeneity in its etiology, pathogenesis, and clinical manifestations depending on the genetic background of individuals.
Environmental Factors
Environmental factors play a significant role in the development of T1DM.
Viral Infections
Viruses such as rubella, mumps, coxsackievirus, encephalomyocarditis virus, and cytomegalovirus are known to be associated with T1DM. Recently, enteroviruses have also attracted attention. Viral infections can damage pancreatic β-cells directly or expose their antigenic components by damaging β-cells, disrupting self-immune tolerance, and triggering autoimmune responses against the β-cells.
Chemical Toxins and Dietary Factors
Animal models of diabetes induced by streptozotocin and alloxan, and cases of human diabetes caused by the rodenticide pyriminil, demonstrate that pancreatic β-cell destruction can result from non-immune-mediated mechanisms (acute injury) or immune-mediated mechanisms (chronic injury at low doses).
Symbiotic Microorganisms
The composition and metabolic products of the gut microbiota and other symbiotic microorganisms are closely associated with the development of T1DM.
The interactions between genetic predisposition and environmental stimuli contribute to the complex etiology and pathogenesis of T1DM, with individual variations significantly influencing how the disease manifests and progresses.
Autoimmunity
Many lines of evidence support the classification of T1DM as an autoimmune disease:
- Genetic susceptibility is closely linked to the HLA region.
- It is often accompanied by other autoimmune diseases.
- Early pathological changes include insulitis.
- Nearly 90% of newly diagnosed T1DM patients have detectable autoantibodies targeting pancreatic β-cells in their serum.
- Immunosuppressive therapy can prevent diabetes induced by low-dose streptozotocin in animal models.
- In monozygotic twins, the twin without diabetes rapidly develops insulitis and pancreatic β-cell destruction after receiving a pancreatic transplant from the twin with diabetes.
Humoral Immunity
Autoantibodies targeting pancreatic β-cells are present in the serum of 90% of newly diagnosed T1DM patients. Important autoantibodies include islet cell antibodies (ICAs), insulin autoantibodies (IAAs), glutamate decarboxylase (GAD65) antibodies, tyrosine phosphatase-like protein antibodies (IA-2A and IA-2BA), and zinc transporter 8 antibodies (ZnT8A). The presence of two or more autoantibody types predicts a 70% likelihood of developing T1DM. Autoantibody testing is valuable for predicting T1DM onset, identifying high-risk individuals, assisting in diabetes classification, and guiding treatment strategies.
Cellular Immunity
Cellular immunity abnormalities play a more crucial role in the pathogenesis of T1DM. These abnormalities involve an imbalance between pathogenic and protective T lymphocytes, with dysregulated interactions among cytokines or other mediators secreted by these cells. The mechanisms are complex and intertwined. The development of T1DM is generally thought to occur in three stages:
- Activation of the immune system.
- Release of various cytokines by immune cells.
- Highly specific autoimmune attacks on pancreatic β-cells, either directly or indirectly mediated by activated T lymphocytes, cytokines, or other mediators, leading to insulitis and β-cell destruction.
Natural History
The progression of T1DM involves the following stages:
Individuals possess genetic susceptibility but exhibit no clinical abnormalities.
Certain environmental triggers, such as viral infections, cause partial pancreatic β-cell destruction and initiate a long-term, chronic autoimmune process. This process is continuous or intermittent and may occur alongside β-cell regeneration. During this stage, various autoantibodies targeting islet cells become detectable. The number of β-cells begins to decline, but glucose tolerance remains normal.
When β-cell damage reaches a critical threshold (typically 10-20% of β-cell function remains in children and adolescents at onset, whereas approximately 40% may remain in adults at onset), insufficient insulin secretion leads to impaired glucose tolerance or clinical diabetes, requiring exogenous insulin therapy.
Pancreatic β-cells are almost completely depleted, resulting in a complete reliance on exogenous insulin for survival.
The natural progression of T1DM varies between individuals. Onset in children or adolescents generally progresses more rapidly, whereas onset in adults tends to progress more slowly. In some cases, it may be clinically challenging to differentiate T1DM from MODY (maturity-onset diabetes of the young) or T2DM.
Type 2 Diabetes Mellitus (T2DM)
T2DM is a polygenic, genetically complex disease caused by the interplay of genetic and environmental factors. As a heterogeneous condition, the etiology and pathogenesis of T2DM remain inadequately understood.
Genetic Factors
The concordance rate for T2DM in monozygotic twins approaches 100%, yet disease onset and progression vary significantly depending on environmental influences. The genetic characteristics of T2DM include:
- Multiple genes are involved, with over 100 susceptibility loci identified globally, each affecting specific aspects of glucose metabolism.
- The contribution of each gene to disease development varies; most are minor-effect genes, but a few may be major-effect genes.
- Each gene only confers a degree of susceptibility, which is neither sufficient nor necessary for disease onset.
- The cumulative effects of multiple genetic abnormalities contribute to overall genetic susceptibility.
- The genetic susceptibility to T2DM exhibits racial and ethnic differences.
Environmental Factors
Environmental influences include aging, modern sedentary lifestyles, overnutrition, insufficient physical activity, prenatal environmental conditions, air pollution, stress, and exposure to chemical toxins.
Pathophysiology
Insulin Resistance and Pancreatic β-Cell Dysfunction
Insulin resistance in tissues (particularly the liver and skeletal muscle) and β-cell dysfunction leading to varying degrees of insulin deficiency are the two principal mechanisms involved in T2DM pathogenesis. The relative importance of these factors differs among individuals and may change during the course of the disease. In the presence of insulin resistance, the pancreatic β-cells may compensate by increasing insulin secretion to maintain normal blood glucose levels. However, when β-cell function fails to compensate, T2DM develops.
Insulin Resistance
This refers to a reduced sensitivity of target organs (mainly the liver, muscle, and adipose tissue) to the actions of insulin. Insulin resistance is a characteristic feature of T2DM and is thought to be the initiating factor in many cases. Genetic factors predisposing to insulin resistance can also influence the compensatory ability of β-cells. The mechanisms underlying insulin resistance in T2DM are not fully understood. Two major hypotheses, lipid overload and inflammation, are currently proposed, and these mechanisms are thought to overlap and interact with each other.
β-Cell Dysfunction
β-cell dysfunction plays a pivotal role in the development of T2DM. The inability of β-cells to compensate for insulin resistance is considered the final common pathway leading to T2DM. During the progression from normal glucose tolerance to impaired glucose regulation (IGR) and then to T2DM, a progressive decline in β-cell function has been observed.
The main manifestations of β-cell dysfunction in T2DM include:
- Defects in Insulin Secretion: In the early stages of T2DM, fasting insulin levels are normal or elevated, and glucose-stimulated insulin secretion is compensatorily increased. However, given the presence of insulin resistance, insulin levels are effectively insufficient. As the disease progresses, the maximal insulin secretory capacity gradually declines to levels below normal.
- Abnormal Insulin Secretion Patterns: These include impaired rapid-phase secretion and disrupted circadian rhythms, specifically defects in pulsatile insulin secretion. After intravenous glucose administration, the first-phase insulin response is diminished or absent, and in oral glucose tolerance tests, early-phase secretion is delayed, diminished, or absent. While the second-phase (or late-phase) insulin secretion may be compensatorily elevated with delayed peaks in early disease stages, further progression leads to reduced responsiveness to both glucose and non-glucose stimuli.
- Qualitative Defects in Insulin Secretion: The ratio of proinsulin to insulin increases, reflecting qualitative abnormalities in insulin secretion.
The causes and underlying susceptibility factors for β-cell dysfunction, as well as the initiating and exacerbating mechanisms of β-cell damage, remain unclear. Multiple factors may be involved, many of which appear to be genetically determined. Potential congenital factors include mitochondrial dysfunction, abnormalities in the tricarboxylic acid cycle, dysregulation of AMP-activated protein kinase (AMPK), and altered uterine or early-life endocrine and nutritional environments, all of which may reduce β-cell quantity. Triggering factors for β-cell damage may include glucolipotoxicity, oxidative stress, and endoplasmic reticulum stress. The progression of β-cell dysfunction may be further aggravated by factors such as islet inflammation, accumulation of advanced glycation end products in the islets, and deposition of fat and/or amyloid in the pancreatic islets. These factors contribute to reduced glucose sensitivity, β-cell dedifferentiation (or transdifferentiation), and/or apoptosis, leading to further structural and functional deterioration.
Moreover, obesity, particularly central obesity, strongly correlates with insulin resistance, β-cell dysfunction, and the development of T2DM.
Abnormal Pancreatic α-Cell Function and Impaired Incretin Secretion
Glucagon plays an important role in the maintenance of glucose homeostasis. Under normal conditions, an increase in postprandial blood glucose levels stimulates pancreatic β-cells to secrete insulin and intestinal L-cells to release glucagon-like peptide-1 (GLP-1), one of the incretins. These processes suppress glucagon secretion from pancreatic α-cells, reducing hepatic glucose output and preventing postprandial hyperglycemia. In T2DM, insufficient insulin secretion, a significant increase in the α/β-cell ratio, reduced sensitivity of α-cells to glucose, and decreased GLP-1 levels result in elevated glucagon secretion and increased hepatic glucose output.
GLP-1 secretion defects also contribute significantly to the pathogenesis of T2DM. GLP-1 stimulates glucose-dependent insulin synthesis and secretion from β-cells, suppresses glucagon secretion, slows gastric emptying, reduces appetite and food intake, promotes β-cell proliferation, decreases apoptosis, improves endothelial function, and protects cardiac function. Studies have demonstrated that the GLP-1 release curve after a glucose load is lower in individuals with T2DM compared to those without diabetes. Increasing GLP-1 levels in T2DM patients has been shown to enhance glucose-dependent insulin secretion, suppress glucagon secretion, and restore glucose sensitivity in α-cells.
Abnormal Function of the Stomach and Small Intestine and Altered Gut Microbiota
Disruption of vagus nerve function may lead to abnormal gastric emptying, increased glucose absorption in the small intestine, and abnormalities in the composition and function of the gut microbiota. These factors may contribute to the development and progression of T2DM.
Abnormal Brain Structure and Function
The brain can influence insulin sensitivity in the body by regulating appetite, mood, cognition, and glucose metabolism in peripheral tissues. Specific brain regions, such as the hypothalamus, contain glucose-sensing systems that detect and respond to circulating glucose levels, playing a critical role in maintaining glucose homeostasis. Structural and functional impairments in these regulatory regions have been observed in individuals with T2DM.
Immune Imbalance and Chronic Inflammation
Immune imbalance and systemic, low-grade chronic inflammation are present in individuals with T2DM. These factors contribute to insulin resistance, increase endoplasmic reticulum stress, and elevate blood glucose levels.
Reduced Renal Glucose Excretion
Individuals with T2DM exhibit an increased renal capacity for glucose reabsorption, leading to an elevated renal glucose threshold.
Natural History
In the early stages of T2DM, insulin resistance is present, but pancreatic β-cells compensate by increasing insulin secretion, maintaining normal blood glucose levels. When β-cells fail to secrete enough insulin to compensate for the insulin resistance, the condition progresses to impaired glucose regulation (IGR) and subsequently to diabetes. In the early stages of IGR and diabetes, some patients can achieve glycemic control through lifestyle interventions alone, while most require oral hypoglycemic agents in addition to lifestyle changes to achieve optimal blood glucose control.
As β-cell insulin secretion progressively declines, some patients require insulin therapy to control hyperglycemia. However, they do not depend on exogenous insulin for survival. With further disease progression, a significant proportion of patients may become reliant on insulin to control blood glucose levels and maintain life.
Clinical Manifestations
Basic Clinical Presentations
Metabolic Disorder Symptoms
Elevated blood glucose levels can lead to osmotic diuresis, resulting in polyuria, which is subsequently followed by thirst and polydipsia. Impaired glucose utilization in peripheral tissues, increased fat breakdown, and negative protein metabolism balance result in fatigue, weight loss, and growth retardation in children. Patients are often excessively hungry and exhibit polyphagia. The clinical manifestations of diabetes are typically described as "three polys and one less," specifically, polyuria, polydipsia, polyphagia, and weight loss. Patients may also experience pruritus, especially vulvar itching. Rapidly rising blood glucose levels can alter the osmotic pressure of the aqueous humor and lens, leading to refractive changes and blurred vision. Many patients remain asymptomatic, with hyperglycemia being detected only during routine health examinations or when seeking medical care for other conditions.
Complications and/or Comorbidities
These are described in subsequent sections.
Clinical Features of Common Types of Diabetes
Type 1 Diabetes Mellitus (T1DM)
The clinical presentation at diagnosis varies widely, ranging from mild, nonspecific symptoms to the typical "three polys and one less" symptoms, or even diabetic coma. Most adolescents with T1DM experience an acute onset with prominent symptoms; delayed diagnosis and treatment may lead to diabetic ketoacidosis (DKA). In the early stages of T1DM, most patients require insulin therapy to restore metabolic balance. However, after this, many patients enter a period lasting several weeks to months during which insulin requirements markedly decrease—a phenomenon known as the "honeymoon phase," attributed to partial recovery of β-cell function. Some adult-onset T1DM patients experience a slow disease onset with mild initial symptoms and may go through a stage during which insulin therapy is not required for varying lengths of time.
Most T1DM patients have plasma basal insulin levels that are lower than normal, with diminished or flat insulin secretion curves following glucose stimulation. At the time of diagnosis, 70%–90% of T1DM patients test positive for autoantibodies against pancreatic β-cells. A subset of patients present with acute onset, experiencing significant reduction or complete depletion of β-cell function, but test negative for these autoantibodies.
Type 2 Diabetes Mellitus (T2DM)
T2DM is a heterogeneous group of disorders that can develop at any age but is more common in adults, typically beginning after the age of 40. The disease often has an insidious onset with relatively mild symptoms, and more than half of patients are asymptomatic. A significant number of cases are first identified through routine health screenings, or after the emergence of chronic complications or comorbid conditions. T2DM frequently has a family history and is rarely associated with spontaneous DKA, though it can occur under stress or during severe infections. Some elderly T2DM patients are diagnosed when presenting with hyperosmolar hyperglycemic syndrome (HHS). T2DM is commonly associated with other conditions such as obesity, dyslipidemia, and hypertension, either concurrently or sequentially.
The clinical course of T2DM varies depending on the stage of disease at the time of diagnosis. In some early-stage, obese patients, plasma basal insulin levels may be normal or elevated, though the peak of insulin secretion in response to glucose stimulation may be delayed. In these cases, plasma insulin levels can rise inappropriately 3 to 5 hours after a meal, inducing reactive hypoglycemia, which may serve as the initial clinical presentation for these patients.
Some Specific Types of Diabetes
Maturity-Onset Diabetes of the Young (MODY)
MODY refers to a group of highly heterogeneous monogenic forms of diabetes. At least 14 subtypes have been identified. The main clinical features include:
- A family history of diabetes spanning at least three generations, following an autosomal dominant inheritance pattern.
- Onset of diabetes before the age of 25.
- A lack of ketosis, with no need for insulin therapy for at least five years post-diagnosis.
Mitochondrial Gene Mutation Diabetes
Clinical features include:
- Maternal inheritance.
- Early onset with progressive decline in β-cell function, and negative autoantibodies.
- A lean body habitus.
- Frequent association with sensorineural deafness or other neuromuscular manifestations.
Steroid-Induced Diabetes
Diabetes can be induced by glucocorticoid use in some individuals. Its occurrence is often related to the dosage and duration of glucocorticoid use, particularly in those already exhibiting insulin resistance or pancreatic β-cell dysfunction. For most patients, glucose metabolism normalizes after discontinuation of glucocorticoids. Blood glucose levels should be monitored in individuals using glucocorticoids, regardless of prior diabetes history, with insulin being the preferred treatment for hyperglycemia during glucocorticoid use.
Gestational Diabetes Mellitus (GDM)
GDM typically develops during the middle or late stages of pregnancy and usually presents as mild, asymptomatic hyperglycemia. Blood glucose levels generally normalize after delivery, though women with GDM have a significantly increased risk of developing T2DM in the future.
Complications
Acute Severe Metabolic Disorders
This category includes diabetic ketoacidosis (DKA) and hyperosmolar hyperglycemic syndrome (HHS), which are discussed in subsequent sections.
Infectious Diseases
Diabetic patients are more susceptible to a wide range of infections, and those with poorly controlled blood glucose are at even greater risk, with infections often being more severe. Pyelonephritis and cystitis are particularly common in female patients, often recurring and, in severe cases, leading to renal or perirenal abscesses and renal papillary necrosis. Skin infections, such as boils and carbuncles, frequently recur and sometimes result in septicemia. Fungal skin infections, such as athlete's foot and tinea corporis, are also common. Women with diabetes often experience complications such as fungal vaginitis or Bartholin’s gland inflammation, typically caused by Candida infections.
The incidence of tuberculosis is significantly higher in individuals with diabetes, and the disease tends to spread more extensively. Imaging findings in these cases are often atypical, making misdiagnosis or delayed diagnosis more likely.
Chronic Complications
Chronic complications in diabetes can affect nearly all tissues and organs in the body. Microvascular disease, atherosclerotic vascular disease, and neuropathy are the fundamental pathological changes underlying chronic diabetic complications. These pathological changes may occur independently or in various combinations, either simultaneously or sequentially.
Chronic complications can sometimes be present even before diabetes is diagnosed, with some patients being diagnosed after their complications prompt further investigation. Diabetes increases the risk of cardiovascular, cerebrovascular, and peripheral vascular diseases by two- to seven-fold. Compared to non-diabetic individuals, people with diabetes have significantly higher risks of all-cause mortality, cardiovascular-related death, and lower limb amputation. Diabetes is the leading cause of adult blindness, non-traumatic amputation, and end-stage renal disease (ESRD). Among these, cardiovascular diseases are the primary cause of disability and death in diabetic patients.
The pathogenesis of chronic diabetic complications is highly complex and remains incompletely understood. It is associated with factors such as genetic susceptibility, insulin resistance, hyperglycemia, chronic low-grade inflammatory states, endothelial dysfunction, and coagulation abnormalities. Hyperglycemia-related vascular damage involves mechanisms such as activation of the polyol pathway, increased formation of advanced glycation end-products (AGEs), activation of the protein kinase C (PKC) pathway, and activation of the hexosamine pathway. Excessive production of reactive oxygen species (ROS) by the mitochondrial electron transport chain during hyperglycemia leads to oxidative stress, which is a unifying mechanism linking these pathways.
Microvascular Complications
Microvascular complications involve damage to tiny blood vessels, specifically capillaries and microvascular networks with luminal diameters less than 100 μm, located between arterioles and venules. Microvascular complications are specific to diabetes, with characteristic features including thickening of the capillary basement membrane and microcirculatory dysfunction.
Microvascular complications can affect almost all tissues and organs but predominantly manifest in the retina, kidneys, nerves, and myocardium. Among these, diabetic kidney disease (DKD) and retinopathy are of particular importance.
Diabetic Kidney Disease (DKD)
DKD refers to chronic kidney disease caused by diabetes. Its pathological changes are complex and involve the glomeruli, renal tubules, and renal interstitial structures.
Glomerular Pathology: Glomerular pathology is a hallmark feature of DKD. Early manifestations include glomerular hypertrophy, thickening of the glomerular capillary basement membrane, and mild mesangial matrix expansion. With disease progression, mesangial expansion further intensifies, and mesangiolysis may occur. In the middle-to-late stages, nodular glomerulosclerosis, known as Kimmelstiel-Wilson (K-W) nodules, develops. These nodules are highly specific to DKD, are linked to diabetes progression, and are associated with poor prognosis. End-stage disease often progresses to diffuse glomerulosclerosis, which is the most common but less specific pathology, as similar changes can occur in mesangiocapillary glomerulonephritis or lupus nephritis. In the advanced stage, exudative lesions may also be observed. Hyalinosis of afferent and efferent arterioles, a feature with diagnostic significance, is often accompanied by arterial intimal thickening.
Tubulointerstitial Lesions: More recently, it has been recognized that tubulointerstitial lesions, such as interstitial fibrosis, tubular atrophy, and interstitial vascular damage, play important roles in the progression of renal dysfunction in DKD. These lesions may even precede glomerular changes. The exact role of these lesions in the pathogenesis and progression of DKD requires further investigation.
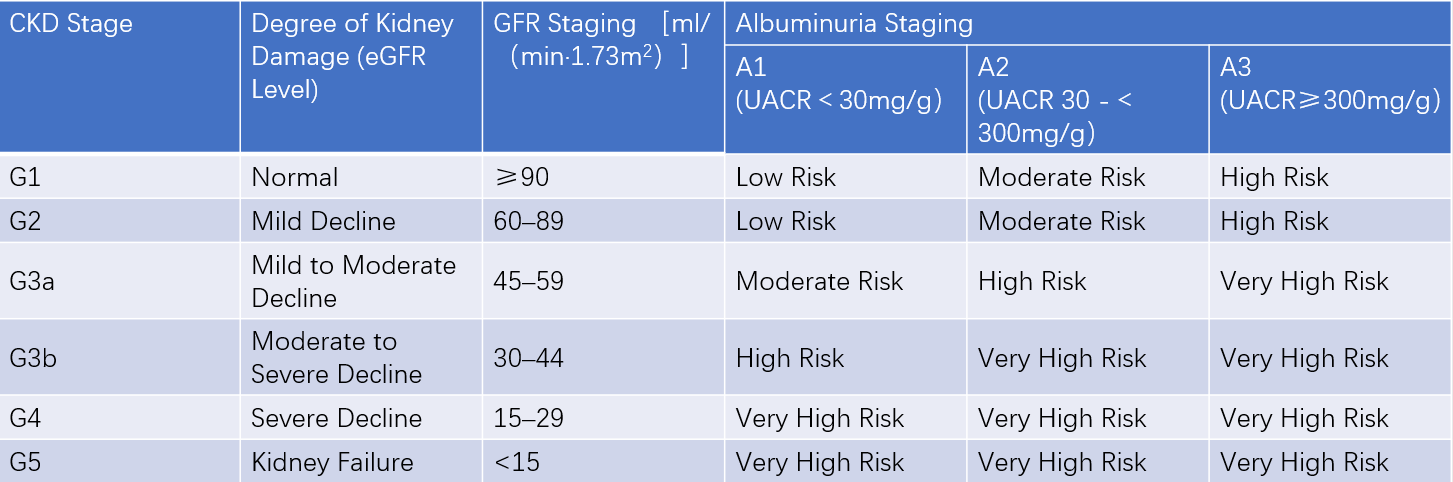
Table 1 CKD staging by eGFR and UACR with progression risk
Clinically, DKD is characterized primarily by persistent proteinuria and/or a progressive decline in the glomerular filtration rate (GFR). Currently, clinical staging of DKD follows the framework of chronic kidney disease (CKD) using combined assessments of estimated glomerular filtration rate (eGFR) and the urine albumin-to-creatinine ratio (UACR), referred to as the GA staging system. In this staging system, "G" represents the level of eGFR and is classified from G1 to G5, while "A" represents the degree of albuminuria, categorized from A1 to A3. The GA staging system is utilized to assess the severity of DKD, predict prognosis, and guide the development of treatment and follow-up strategies.
DKD is a leading cause of end-stage renal disease (ESRD) and the primary cause of death in type 1 diabetes mellitus (T1DM). In type 2 diabetes mellitus (T2DM), it ranks second only to cardiovascular and cerebrovascular diseases in terms of severity. DKD is an important subtype of CKD, and diabetic patients may develop renal microvascular complications, frequently alongside other conditions such as hypertension, dyslipidemia, atherosclerosis, and/or other chronic kidney diseases. These factors collectively contribute to the onset and progression of CKD in diabetic individuals, and in most cases, the involvement of multiple factors makes it clinically challenging to delineate the exact causes. Pathological examination holds significant value in distinguishing the etiology of chronic renal impairment and is considered the "gold standard" for diagnosing DKD (specifically diabetic glomerulopathy, or DG) and differentiating it from non-diabetic kidney disease (NDKD) in cases where clinical diagnosis is difficult. However, histological changes observed in renal biopsies do not consistently correlate with the clinical manifestations or the degree of renal functional impairment.
Diabetic Retinopathy (DR)
Diabetic retinopathy refers to a series of lesions caused by microvascular disease in the retina and is a specific complication of diabetes. Among patients with more than 10 years of diabetes duration, varying degrees of DR are frequently observed. It is a primary cause of blindness due to diabetes. DR is currently classified into two major categories and six stages:
- Stage I (Mild Non-Proliferative DR): Characterized by microaneurysms.
- Stage II (Moderate Non-Proliferative DR): Includes retinal hemorrhages, hard exudates, and/or cotton-wool spots.
- Stage III (Severe Non-Proliferative DR): Defined by any of the following: ≥20 intraretinal hemorrhages in each of four quadrants, significant venous beading in at least two quadrants, or intraretinal microvascular abnormalities in at least one quadrant. DR in this stage is termed "very severe non-proliferative DR" if two or more of these features are present.
- Stage IV (Early Proliferative DR): Newly formed blood vessels are seen on the retina or optic disc.
- Stage V (Fibrovascular Proliferative Stage): Fibrovascular membranes develop, often accompanied by preretinal hemorrhages or vitreous hemorrhages.
- Stage VI (Late Proliferative Stage): Characterized by tractional retinal detachment, which may coexist with fibrovascular membranes, preretinal hemorrhages, or vitreous hemorrhages.
Stages I–III are collectively referred to as non-proliferative diabetic retinopathy (NPDR), whereas stages IV–VI are considered proliferative diabetic retinopathy (PDR). Diabetic macular edema (DME), defined as retinal thickening caused by capillary leakage in the macular region, mainly affects central vision. PDR often coexists with DKD and neuropathic complications.
Other Complications
When structural and functional changes in the heart attributable to diabetes are not associated with coronary artery disease or hypertension, the term diabetic cardiomyopathy is used. This condition may result from microvascular damage in the heart and metabolic disturbances in myocardial tissue. It may lead to heart failure, arrhythmias, cardiogenic shock, or sudden cardiac death and may coexist with other cardiac conditions.
Atherosclerotic Cardiovascular Disease (ASCVD)
The prevalence of risk factors for atherosclerosis, such as obesity, hypertension, and dyslipidemia, is significantly higher among individuals with diabetes (especially T2DM), resulting in earlier onset, higher prevalence, and faster progression of atherosclerosis within this population. Atherosclerosis commonly affects the aorta, coronary arteries, cerebral arteries, renal arteries, and peripheral arteries, leading to coronary artery disease, ischemic or hemorrhagic cerebrovascular disease, renal artery sclerosis, and peripheral artery sclerosis. Cardiovascular and cerebrovascular diseases remain the leading causes of death in diabetic patients.
Neurological Complications
Neurological complications are the most common chronic complications of diabetes and can affect any part of the nervous system. The etiology is complex, potentially involving ASCVD and microvascular disease, metabolic factors, autoimmune mechanisms, and deficiencies in growth factors.
Central Nervous System Complications
These include:
- Altered mental status associated with severe DKA, HHS, or hypoglycemia.
- Stroke.
- Accelerated brain aging and conditions such as Alzheimer's disease.
Diabetic Peripheral Neuropathy
Common types of diabetic peripheral neuropathy include:
Diffuse Neuropathy
Distal symmetric polyneuropathy is the most common form, typically involving sensory and motor nerves in the distal extremities. It is usually symmetrical and is characterized by a "glove-and-stocking" distribution. Symptoms are more pronounced and occur earlier in the lower limbs compared to the upper limbs, often presenting as sensory abnormalities in the extremities, which may be accompanied by hyperalgesia or pain. In advanced stages, patients may experience sensory loss and involvement of motor nerves, leading to atrophy of small muscles in the hands and feet, sensory ataxia, and neurogenic arthropathy (Charcot joints). Reflexes are typically hyperactive in the early stages but become diminished or absent in later stages, while vibratory sensation on tuning fork examination is reduced or lost. Electrophysiological studies can detect early slowing of sensory and motor nerve conduction velocities.
Autonomic neuropathy often affects the gastrointestinal, cardiovascular, and genitourinary systems. Clinical manifestations include delayed gastric emptying (gastroparesis), diarrhea (postprandial or nocturnal), and constipation. Cardiovascular symptoms include orthostatic hypotension, silent myocardial infarction, and QT interval prolongation, with severe cases potentially resulting in sudden cardiac death. Urological symptoms include increased post-void residual volume, urinary incontinence, and urinary retention. Other possible features include erectile dysfunction, pupillary abnormalities, and alterations in sweating patterns.
Mononeuropathy
This form may affect any cranial or spinal nerve, with the oculomotor, median, and peroneal nerves being the most commonly involved. The onset is typically acute, presenting as pain in the distribution of the affected nerve, and tends to be self-limiting. Neuropathy involving multiple discrete nerves is referred to as mononeuritis multiplex, which needs to be differentiated from polyneuropathy.
Radiculopathy or Plexopathy
The most common presentation is lumbar radiculopathy, typically manifesting as initial pain in the hip, thigh, or buttocks, which progresses to weakness and atrophy of proximal pelvic muscles.
The diagnosis of diabetic peripheral neuropathy requires exclusion of other causes of neuropathy.
Diabetic Foot
Diabetic foot refers to foot ulcers, infections, and/or deep tissue destruction associated with distal neuropathy of the lower extremities and varying degrees of peripheral vascular disease. It is one of the most severe and costly chronic complications of diabetes and the leading cause of non-traumatic lower-extremity amputations in diabetic patients. Mild presentations include foot deformities, dry or cool skin, and the formation of calluses. Severe cases may involve foot ulcers and gangrene, which can lead to amputation and death.
Other Diabetes-Related Complications
Diabetes can also result in additional complications, such as cataracts, glaucoma, and iris or ciliary body disorders. Oral diseases are common among individuals with diabetes, and abnormalities in glucose metabolism are frequently observed in patients with oral diseases. Certain skin disorders are specific to diabetes, although most are non-specific. Additionally, the incidence of cancers such as liver, pancreatic, and bladder cancer is increased in diabetic patients. Mental health conditions, including depression, anxiety, and cognitive impairment, are also more prevalent in this population.
Laboratory Tests
Evaluation of Abnormal Glucose Metabolism or its Control
Urine Glucose Testing
Positive urine glucose tests are an important clue in the diagnosis of diabetes. However, a positive result only indicates that blood glucose levels have exceeded the renal threshold (approximately 10 mmol/L), and a negative result does not exclude the possibility of diabetes.
Blood Glucose Testing and Oral Glucose Tolerance Test (OGTT)
Blood glucose values reflect the instantaneous blood glucose state. Testing can be performed using venous blood or capillary blood to measure plasma, serum, or whole blood glucose levels. If the hematocrit is within normal range, plasma and serum glucose levels are approximately 15% higher than whole blood glucose levels. For diagnosing diabetes, venous blood should be used to measure blood glucose, while portable glucose meters can be used for follow-up monitoring of peripheral blood glucose during treatment.
If blood glucose levels are above the normal range but do not meet the diagnostic criteria for diabetes, an OGTT may be indicated. OGTT should be performed in the morning after an 8-hour period of fasting with no caloric intake. For adults, 75 g of anhydrous glucose is dissolved in 250–300 mL of water and consumed within 5–10 minutes. Venous blood glucose levels are measured at fasting baseline and at 2 hours post glucose ingestion. For children, the glucose dose is calculated based on 1.75 g/kg of body weight, with a maximum dose of 75 g.
Several factors can affect the accuracy of OGTT results, including inadequate carbohydrate intake for three consecutive days prior to the test, prolonged bed rest or minimal physical activity, stress, or the use of certain medications (e.g., thiazide diuretics, glucocorticoids). Therefore, OGTT is not recommended during acute illness or stress. It is advised to ensure adequate carbohydrate intake for at least three days prior to testing and to discontinue any medications that could affect results 3–7 days before the test. During the test, participants are advised to avoid tea, coffee, smoking, and strenuous exercise in order to minimize confounding factors.
Glycated Hemoglobin (GHbA1) and Glycated Albumin (GA) Measurements
Glycated hemoglobin (GHbA1) is a product of a non-enzymatic glycation reaction between glucose (or other sugars) and the amino groups of hemoglobin. Its levels are positively correlated with blood glucose concentrations. GHbA1 has three subtypes: a, b, and c, among which GHbA1c (HbA1c) is the most clinically significant. In healthy individuals, HbA1c accounts for 3%–6% of total hemoglobin, although reference ranges may vary between laboratories. HbA1c is an important indicator used in the diagnosis of diabetes and for assessing blood glucose control. Elevated HbA1c levels are observed in individuals with poor glycemic control and are associated with both the degree and duration of hyperglycemia. Since the average lifespan of red blood cells in circulation is approximately 120 days, HbA1c reflects the patient’s average blood glucose levels over the past 8–12 weeks. However, several factors can affect HbA1c levels, including the testing method, the presence of anemia or hemoglobinopathies, red blood cell turnover rates, and age. HbA1c cannot reflect real-time glucose levels, glucose fluctuations, or the occurrence of hypoglycemia.
Similarly, serum albumin can undergo a non-enzymatic glycation reaction with glucose to form glycated albumin (GA). The amount of GA formed is also related to the concentration and duration of hyperglycemia, with a normal range of 11%–17%. Since albumin has a half-life of approximately 19 days in the bloodstream, GA reflects the patient’s average blood glucose levels over the past 2–3 weeks. GA is a useful indicator for evaluating short-term glycemic control in diabetic patients. However, certain conditions that affect albumin turnover (e.g., nephrotic syndrome, liver cirrhosis) may impact GA measurements.
Self-Monitoring of Blood Glucose (SMBG)
Self-monitoring of blood glucose involves measuring capillary blood glucose (BGM) using portable glucometers or continuous glucose monitoring (CGM) systems. CGM uses glucose sensors to continuously monitor changes in interstitial glucose concentrations in the subcutaneous tissue.
Pancreatic β-Cell Function Tests
Insulin Secretion Test
In healthy individuals, fasting plasma insulin levels range from 35 to 145 pmol/L (5–20 mU/L). Following oral administration of 75 g of anhydrous glucose (or steamed bread made from 100 g of standard flour), plasma insulin typically peaks within 30–60 minutes, with a peak value 5–10 times the baseline. Plasma insulin levels then return to baseline within 3–4 hours. This test reflects basal and glucose-mediated insulin secretion.
C-Peptide Secretion Test
The procedure is similar to the insulin secretion test. In healthy individuals, the fasting baseline C-peptide level is no less than 400 pmol/L, with a peak occurring at the same time as insulin levels and reaching a value 5–6 times the baseline. This test also reflects basal and glucose-mediated insulin secretion.
Other Methods for Assessing β-Cell Function
Intravenous Glucose Tolerance Test (IVGTT)
This provides information about the first-phase insulin response to glucose stimulation.
Hyperglycemic Clamp Test
This measures glucose-mediated first-phase insulin release.
Glucagon-Stimulated C-Peptide Test and Arginine-Stimulated C-Peptide Test
These assess non-glucose-mediated insulin secretion capacity.
Evaluation of Insulin Resistance
Currently, there is no universally accepted clinical standard for assessing insulin resistance.
Simple anthropometric indices (e.g., body mass index, waist circumference, waist-to-hip ratio) and blood insulin levels can be used clinically for a general assessment of insulin resistance and its severity.
The hyperinsulinemic-euglycemic clamp is the internationally recognized "gold standard" for evaluating insulin resistance, although it is not practical for routine clinical use.
Simple and efficient methods, such as the homeostasis model assessment of insulin resistance (HOMA-IR) and the quantitative insulin sensitivity check index (QUICKI), are commonly used. However, the appropriateness of each method depends on the population being investigated and the factors influencing results.
Tests Related to Etiology and Pathogenesis
Combined testing for GAD65 antibodies, islet cell antibodies (ICA), insulin autoantibodies (IAA), IA-2A, and ZnT8A is used for detailed assessment. Genetic analysis can also provide additional insights.
Complications Screening
Screening for complications includes assessments for acute severe metabolic disturbances, such as ketone bodies, electrolytes, and acid-base imbalances. Other evaluations include assessments of the cardiovascular, hepatic, renal, cerebral, ocular, oral, vascular, and nervous systems.
Diagnosis and Differential Diagnosis
Diabetes mellitus should be identified and diagnosed as early as possible in clinical practice to facilitate timely treatment. The diagnosis of diabetes is based on elevated blood glucose levels. The normal range and cutoff values for abnormal glucose metabolism are determined according to the relationship between blood glucose levels and the risk of diabetes-specific complications, such as retinopathy. The use of fasting plasma glucose (FPG) alone is associated with a high rate of missed diagnoses. When necessary, postprandial blood glucose or an oral glucose tolerance test (OGTT) should also be measured. Diagnoses should consider whether they meet the criteria for diabetes, the classification of diabetes, the presence and severity of complications, and the existence of concomitant conditions or factors exacerbating diabetes.
Diagnostic Clues
Diagnostic clues include:
- Symptoms of polyuria, polydipsia, polyphagia, and weight loss ("three polys and one loss").
- Initial presentation with acute or chronic complications of diabetes or related comorbidities.
- High-risk individuals, such as those with a history of impaired glucose regulation (IGR), individuals aged ≥ 35 years, overweight or obese individuals, first-degree relatives of patients with type 2 diabetes (T2DM), those with a history of gestational diabetes mellitus (GDM), individuals with a sedentary lifestyle, those with polycystic ovary syndrome, or individuals receiving long-term antidepressant or glucocorticoid therapy.
In addition, routine diabetes screening is recommended for individuals over 35 years of age undergoing health check-ups or those hospitalized for other illnesses or surgeries.
Diagnostic Criteria
The current diagnostic and classification criteria for diabetes are internationally standardized.
Blood glucose assessments used in the diagnosis of diabetes include fasting plasma glucose (FPG), random blood glucose (measured at any time of day), 2-hour plasma glucose (2hPG) during an OGTT, or glycated hemoglobin (HbA1c). Fasting is defined as no caloric intake for at least 8 hours. Random blood glucose refers to measurements taken at any time of the day, irrespective of the time since the last meal or the quantity of food consumed. Random blood glucose cannot be used to diagnose impaired fasting glucose (IFG) or impaired glucose tolerance (IGT).
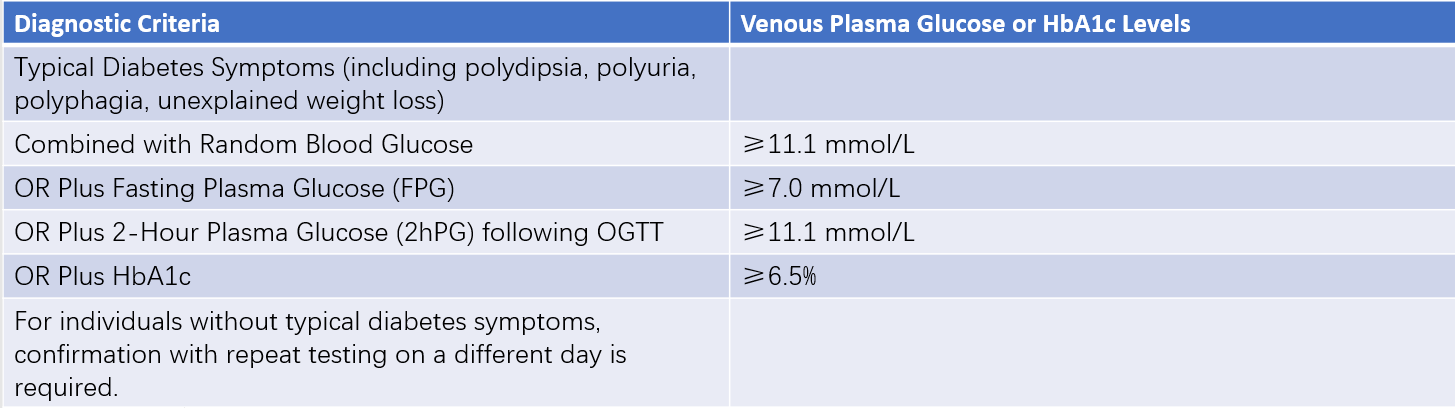
Table 2 Diagnostic criteria for diabetes

Table 3 Classification of glucose metabolism status (based on WHO diabetes expert committee report)
Note: The International Diabetes Expert Committee recommends an IFG threshold level of 5.6–6.9 mmol/L.
Stress hyperglycemia may occur in cases of severe illness or stress, presenting as a temporary and self-limited metabolic disturbance. In these cases, diabetes cannot be diagnosed based on blood glucose levels recorded during the stressful state. Re-evaluation of glucose metabolism is required once the stress has subsided.
Diagnostic criteria for children with diabetes are the same as those for adults.
Gestational diabetes mellitus (GDM) refers to diabetes first identified during pregnancy through antenatal screening. Women diagnosed with type 1 diabetes (T1DM) or T2DM prior to pregnancy are not classified under GDM. For women with normal glucose levels during initial screening, a 75 g OGTT should be performed at 24–28 weeks of gestation if any of the following criteria are met: FPG ≥ 5.1 mmol/L, 1hPG ≥ 10.0 mmol/L, and/or 2hPG ≥ 8.5 mmol/L.
HbA1c is an important marker for evaluating long-term glucose control and guiding adjustments to treatment in diabetic patients. With the gradual standardization of HbA1c testing, an HbA1c threshold of ≥ 6.5% is used as a complementary diagnostic criterion for diabetes. If factors are present that affect HbA1c measurement, alternative testing methods without interference or venous blood glucose levels are used to confirm the diagnosis.
Differential Diagnosis
Distinction from other conditions causing glycosuria is important for differential diagnosis.
Classification
Diabetes mellitus is a highly heterogeneous group of conditions. The 2019 WHO classification update aimed to provide a globally applicable framework to guide clinical diagnosis and the selection of appropriate treatment strategies.
According to the current classification, type 1 diabetes (T1DM) and type 2 diabetes (T2DM) are the two major forms of diabetes. Since T1DM and T2DM lack definitive clinical, biochemical, or genetic markers, differentiation primarily relies on clinical features and disease progression. This includes considerations such as age at onset, speed of onset, severity of symptoms, body weight, the propensity for diabetic ketoacidosis (DKA), and dependence on exogenous insulin for survival. Comprehensive analysis involving β-cell autoantibodies and pancreatic β-cell function testing is also used for diagnosis. Importantly, the differences between T1DM and T2DM are relative rather than absolute. In some cases, patients exhibit features of both T1DM and T2DM, which are classified as "mixed diabetes."
Additionally, "monogenic diabetes" and various forms of "secondary diabetes" belong to the category of "other specific types of diabetes," as their etiology is relatively clear, and some patients may benefit from precision treatments.
In certain cases, the classification of diabetes may not be clear at the time of diagnosis. Such cases are temporarily categorized as "unclassified diabetes," with future classification determined based on initial response to treatment, dynamic changes in β-cell function, and genetic testing.
Diagnosis of Complications and Comorbidities
Appropriate evaluations should be performed for complications of diabetes and associated conditions such as obesity, hypertension, dyslipidemia, fatty liver, obstructive sleep apnea, malignancies, cognitive impairment, anxiety disorders, and depression to facilitate timely diagnosis and treatment.
For T1DM, autoimmune disease screenings should be performed based on presenting symptoms and signs.
Management
The short-term goal of diabetes management focuses on controlling hyperglycemia and associated metabolic disturbances to relieve symptoms, prevent acute severe metabolic complications, and achieve remission in type 2 diabetes mellitus (T2DM) where possible. The long-term goal is to prevent and/or delay the onset and progression of chronic complications, maintain health and the ability to study or work, support normal growth and development in children, improve quality of life, reduce mortality, and strive for a normal or near-normal life expectancy.
With the growing and deepening understanding of diabetes pathophysiology, the concept of diabetes management has evolved. There is an increasing emphasis on patient self-management, education, and support as well as a multidisciplinary, patient-centered collaborative care model. Management strategies prioritize both prevention and treatment, with glucose control tailored toward the prevention and management of complications. Long-term and rational control of multiple cardiovascular risk factors is emphasized to achieve comprehensive goals. Individualized treatment plans are developed based on the clinical characteristics of the patient, taking into consideration their preferences and needs.
Comprehensive medical assessments are essential in diabetes management and include evaluations of patient self-care capabilities, the presence of complications and comorbidities, nutritional status, psychosocial condition, and social support systems. Such assessments should be performed during the initial visit and periodically during follow-up.
Sustained good glycemic control can delay the onset and progression of microvascular complications in newly diagnosed diabetic patients. Early and effective glucose control may also provide long-term cardiovascular protection (a phenomenon known as the "metabolic memory" effect), preserve pancreatic β-cell function, and improve insulin sensitivity. Comprehensive management of multiple risk factors can significantly reduce the occurrence and mortality risks of both microvascular complications and atherosclerotic cardiovascular disease (ASCVD). The International Diabetes Federation (IDF) identifies five key elements of comprehensive diabetes management, known as the "Five Horsemen": diabetes education, medical nutrition therapy, exercise therapy, blood glucose monitoring, and pharmacological treatment.
Management Strategies and Treatment Pathways for Hyperglycemia in Diabetes
Diabetes mellitus is a highly heterogeneous group of diseases, and individualized treatment is realistically feasible for only a subset of monogenic or secondary forms of diabetes. Type 1 diabetes mellitus (T1DM) is characterized by absolute insulin deficiency, and insulin injection is the fundamental treatment option. For T2DM, genetic and environmental factors play varying roles in individual susceptibility, and treatment regimens are primarily designed based on clinical features. Future advancements aim to subclassify T2DM into distinct subtypes based on clinical characteristics, genomics, pathophysiology, and other omic technologies to develop tailored precision management strategies.
Achieving and maintaining appropriate body weight is central to the prevention and management of obesity-related and overweight-associated T2DM. A healthy lifestyle remains an integral part of diabetes treatment.
Blood glucose control is crucial in the comprehensive management of diabetes. Individualized management plans, glucose-lowering regimens, and HbA1c targets should be developed for T2DM patients, taking into account factors such as disease characteristics, economic status, educational background, treatment adherence, and healthcare resources.
For T2DM patients without ASCVD, high ASCVD risk, or chronic kidney disease (CKD), the primary therapeutic goals include achieving and maintaining blood glucose and weight targets. The choice of glucose-lowering strategies and medications should comprehensively account for glucose-lowering goals, issues related to overweight and obesity, risk of hypoglycemia, accessibility, and cost. Metformin is generally recommended for early use due to its effective glucose-lowering potential, additional non-glycemic benefits, favorable cost-effectiveness ratio, and widespread availability. For newly diagnosed patients with significantly elevated baseline blood glucose levels (e.g., HbA1c ≥ 7.5%), simultaneous initiation of two glucose-lowering agents may be considered. For patients with extremely high baseline glucose levels (e.g., HbA1c ≥ 9.0% or FPG ≥ 11.1 mmol/L), short-term intensive insulin therapy may be initiated directly. When dual therapy fails to achieve glycemic targets, a third glucose-lowering drug may be added. If blood glucose levels remain uncontrolled, treatment may be escalated to include multiple daily subcutaneous insulin injections or continuous subcutaneous insulin infusion (CSII, also known as insulin pump therapy).
For T2DM patients with ASCVD or high ASCVD risk, heart failure, or CKD, the treatment focus is on slowing the progression of chronic complications, reducing the risk of cardiovascular, cerebrovascular, and renal events, and improving overall outcomes. Comprehensive control of ASCVD risk factors and maintaining long-term stable glycemic control are emphasized. Even for patients whose HbA1c levels are at target, the inclusion of GLP-1 receptor agonists (GLP-1RAs) and/or sodium-glucose co-transporter-2 inhibitors (SGLT-2is) with proven cardiovascular and renal benefits is recommended in treatment plans, provided there are no contraindications.
For most non-pregnant adults, a reasonable HbA1c target is < 7%. Stricter targets (e.g., ≤ 6.5%) may be appropriate for patients with short disease duration, long life expectancy, and without significant cardiovascular disease. Conversely, more lenient targets (e.g., ≥ 8.0%) may be appropriate for patients with a history of severe hypoglycemia, limited life expectancy, significant microvascular complications, or established ASCVD.
Ongoing follow-up is essential, with regular evaluations of treatment regimens and medication adherence every 3–6 months. Efforts should aim for safe and stable glycemic control that meets comprehensive goals both in the short and long term.
Health Education
Health education for diabetes is a fundamental management measure and plays a key role in the success of diabetes care. It includes training and continuing medical education for diabetes prevention and treatment professionals, as well as education for patients, their families, and the general public on diabetes prevention and management. Diabetes self-management education and support (DSMES) focuses on patient-centered care, helping individuals acquire knowledge and skills for managing their disease. The use of new auxiliary learning technologies, such as the internet and mobile applications, is encouraged.
Medical Nutrition Therapy (MNT)
Medical nutrition therapy is a foundational element of comprehensive management. Its primary goals are to help establish healthy eating habits, achieve good metabolic control, reduce risk factors for ASCVD, improve health conditions, and achieve and maintain a healthy body weight. Nutritionists develop individualized MNT plans for patients, determining total caloric intake and distributing nutrients in a balanced manner with scientific meal planning.
Rational Control of Total Calories
Individuals below ideal body weight, children, pregnant or lactating women, or those with wasting conditions may increase caloric intake by 10%–20%. Calories for obese patients may be moderately reduced to gradually return body weight to approximately ±5% of ideal weight. Daily caloric requirements depend on ideal body weight and activity levels. Ideal body weight is calculated as:
Ideal body weight (kg) = Height (cm) - 105.
For adults with normal body weight:
- Completely bedbound individuals: 15–20 kcal per kg of ideal weight per day.
- Resting state: 25–30 kcal per kg of ideal weight per day.
- Light physical activity: 30–35 kcal per kg of ideal weight per day.
- Moderate physical activity: 35–40 kcal per kg of ideal weight per day.
- Heavy physical activity: over 40 kcal per kg of ideal weight per day.
Long-term nutritional therapy with very low energy intake (<800 kcal/day) is not recommended for diabetic patients.
Nutrient Distribution
Carbohydrates should account for 50%–60% of the total caloric intake. Different types of carbohydrates have varying impacts on blood glucose levels, which can be assessed using the glycemic index (GI). The GI reflects the physiological blood glucose response to consumption of specific foods:
- GI ≤ 55%: Low-GI foods.
- 55% < GI < 70%: Medium-GI foods.
- GI ≥ 70%: High-GI foods.
Patients with diabetes should prioritize low-GI foods. Intake of mono- and disaccharides should be restricted, with moderate consumption of sugar alcohols and non-nutritive sweeteners.
Protein intake should constitute 15%–20% of total calories. Adult patients are recommended to consume 0.8–1.2 g of protein per kg of body weight daily. Special populations, such as pregnant or lactating women, malnourished individuals, or those with wasting diseases, may require 1.5–2.0 g/kg. Patients with overt proteinuria are advised to limit protein intake to 0.8 g/kg, while those with declining GFR may require as low as 0.6 g/kg, supplemented with compound α-keto acid preparations. At least half of the protein should come from animal sources to ensure an adequate supply of essential amino acids.
Fat intake should account for 20%–30% of total calories, with saturated fat accounting for less than 10% of total caloric intake, and cholesterol intake limited to less than 300 mg/day.
Foods rich in dietary fiber help slow digestion, reduce postprandial glucose spikes, and promote satiety. Adults are recommended to consume 25–30 g of dietary fiber per day. Daily salt intake should be limited to less than 5 g. Smoking cessation and alcohol limitation are advised.
Rational Meal Distribution
After determining total daily caloric requirements and nutrient distribution (carbohydrates, proteins, and fats), calories should be converted into food equivalents to develop individualized meal plans. Meal planning should align with personal living habits, disease conditions, and medication requirements, ensuring regular mealtimes and fixed portions. Attention should be given to meal sequencing.
Follow-Up
Regular follow-up is essential to adjust nutritional therapy plans according to changes in the patient’s condition.
Exercise Therapy
Exercise is an integral part of diabetes management, particularly for overweight or obese T2DM patients. Physical activity helps control blood glucose and manage body weight. Under medical guidance, patients may engage in regular, appropriate exercises, gradually increase intensity, and maintain long-term adherence. Sedentary periods lasting more than 30 minutes should be interrupted with brief physical activity. T2DM adults are recommended to perform at least 150 minutes of moderate-intensity aerobic exercise and 2–3 sessions of resistance exercise per week.
Blood glucose should be monitored before and after exercise. For high-intensity or prolonged exercise, adjustments in food intake or medication may be needed to prevent hypoglycemia. Exercise in patients with T1DM is best performed postprandially to avoid large fluctuations in blood glucose. Exercise is temporarily unsuitable during acute complications, severe chronic complications, or when there is significant blood glucose variability, frequent recent hypoglycemia, or blood glucose >14–16 mmol/L.
Monitoring Disease Progression
Disease monitoring includes blood glucose assessment, surveillance of ASCVD risk factors, and monitoring for complications.
Basic blood glucose monitoring involves fasting glucose, postprandial glucose, and HbA1c measurements. Patients are encouraged to perform self-monitoring of blood glucose (SMBG), which aids in tracking glucose control and guiding treatment adjustments. Continuous glucose monitoring (CGM) provides more comprehensive data on glucose trends, including daily fluctuations and detection of asymptomatic hypoglycemia.
Time in range (TIR), calculated from CGM data, serves as an effective indicator of glycemic control and treatment decision-making. TIR measures the duration or percentage of time within the target glucose range (typically 3.9–10.0 mmol/L) over 24 hours, with a recommended control target of over 70%, though this may vary with individual conditions.
HbA1c reflects long-term blood glucose control and serves as a critical parameter for treatment evaluation and adjustment. Patients should undergo HbA1c testing at diagnosis, every three months during treatment initiation, and at least twice a year once glycemic targets are achieved. Glycated albumin (GA) can also be used to assess glycemic control over the past 2–3 weeks.
For patients with prediabetes or diabetes, blood pressure should be measured at each visit. Comprehensive assessments of lipids, cardiovascular health, kidney function, neurological status, and eye health should be conducted at least annually to ensure timely intervention when necessary.
Pharmacological Treatment of Hyperglycemia
Pharmacological treatment for hyperglycemia includes two main categories: oral medications and injectable formulations. When dietary and exercise interventions fail to achieve glycemic targets, glucose-lowering medications are introduced in a timely manner.
Oral Glucose-Lowering Medications
Type 2 diabetes mellitus (T2DM) is a progressive disease. For the majority of patients, achieving adequate glycemic control requires pharmacological therapy, often involving a combination of multiple oral glucose-lowering medications.
Biguanides
Metformin, a widely used biguanide, is considered the cornerstone medication for T2DM treatment.
Metformin exerts its metabolic regulatory effects primarily through activation of the AMPK signaling pathway, leading to reduced hepatic glucose production, improved peripheral insulin sensitivity, reduced intestinal glucose absorption, and increased GLP-1 levels. These mechanisms contribute to blood glucose reduction. Additional benefits include improvement of lipid profiles, increased fibrinolytic activity, reduced platelet aggregation, and inhibition of smooth muscle cell and fibroblast proliferation in arterial walls, which may help prevent or alleviate vascular complications associated with diabetes. Metformin can lower HbA1c by 1%–2% and does not promote weight gain.
Indications:
- First-line medication for T2DM treatment, suitable as monotherapy or in combination with other glucose-lowering drugs.
- Approved for monotherapy or combination therapy with insulin in pediatric and adolescent patients aged 10 years and older.
Contraindications and Special Considerations:
- Contraindicated when eGFR < 30 ml/min. For eGFR between 30 and 45 ml/min, benefit-risk evaluations are required, and if metformin is used, dosage adjustments should be made.
- Not recommended for patients with T2DM presenting acute severe metabolic disturbances, liver dysfunction, severe infections, hypoxia, trauma, major surgery, pregnancy, or lactation. Similarly, not suitable for patients with chronic gastrointestinal diseases or malnourishment.
- Should not be used as monotherapy in T1DM.
- Contraindicated in individuals with a history of allergic reactions or severe adverse responses to the medication.
- Should be avoided in individuals with chronic alcohol abuse.
Adverse Reactions:
- Gastrointestinal Reactions: These are the most common side effects. Gastrointestinal symptoms can be minimized by taking the medication with meals, starting with a low dose, and gradually increasing the dosage.
- Skin Allergic Reactions: Rarely reported.
- Lactic Acidosis: This is a serious but rare adverse effect and requires strict adherence to recommended dosing guidelines.
- Hypoglycemia Risk: Metformin rarely causes hypoglycemia when used alone but may increase the risk when combined with insulin or insulin secretagogues.
- Vitamin B12 Deficiency: Long-term use may lead to vitamin B12 deficiency.
Clinical Application
The optimal effective dose of metformin is 2,000 mg/day, with a maximum recommended dose of 2,550 mg/day for adults. For patients with eGFR > 60 ml/min, metformin should be discontinued during intravenous administration of iodine-based contrast agents. For patients with eGFR 45–60 ml/min, metformin should be discontinued 48 hours before imaging procedures involving contrast agents. All patients can resume metformin use 48 hours after the procedure if renal function tests show no deterioration.
Insulin Secretagogues
Sulfonylureas (SU)
Sulfonylureas stimulate insulin secretion from pancreatic β-cells, with their insulinotropic effects being independent of blood glucose levels. The effectiveness of SU medications relies on the presence of a sufficient number of functional β-cells. SUs can reduce HbA1c by 1%–2%. Commonly used sulfonylureas include glibenclamide, glipizide (and glipizide extended-release tablets), gliclazide (and gliclazide sustained-release tablets), glibornuride, and glimepiride.
Indications:
- Used as monotherapy for newly diagnosed T2DM patients who are non-obese and have suboptimal glycemic control with diet and exercise. As T2DM progresses, SUs are often combined with other glucose-lowering medications or insulin to improve glycemic control. SUs lose effectiveness in late-stage T2DM when β-cell function has significantly declined.
Contraindications or Special Considerations:
- Not suitable for patients with T1DM, severe complications, or significantly impaired β-cell function in T2DM. Contraindicated in children and adolescents, pregnant or lactating women, those in the perioperative period of major surgery, post-total pancreatectomy, and individuals allergic to SUs or who have experienced severe adverse effects.
Adverse Reactions:
- Hypoglycemia: The most common and significant side effect, particularly in elderly patients, those with liver or kidney dysfunction, excessive dosage, irregular or reduced food intake, or excessive physical activity.
- Weight Gain: A notable side effect.
- Skin Allergic Reactions: Occasionally observed.
- Hepatic Effects: Rare cases of liver dysfunction or cholestatic jaundice.
- Cardiovascular Effects: Some SUs may reduce myocardial ischemic preconditioning, although current evidence does not suggest that they increase the incidence or mortality of cardiovascular disease in T2DM patients.
Clinical Application:
- Treatment should begin with a low dose, taken 30 minutes before breakfast, with gradual increases in dosage. For higher doses, the medication can be split into two doses taken before breakfast and dinner. Glibenclamide is potent but has a higher risk of hypoglycemia and requires caution in elderly patients and those with liver, kidney, or cardiac dysfunction. Glipizide, gliclazide, and glibornuride have milder effects, making them more suitable for elderly patients. Mild renal impairment does not preclude the use of SUs, but for moderate impairment, glibornuride is preferred, while it is not recommended for severe renal impairment. Combining two SUs or combining SUs with other insulin secretagogues is not recommended.
Glinides
Non-sulfonylurea insulin secretagogues, also known as glinides, lower postprandial blood glucose by stimulating early-phase insulin secretion. They are characterized by rapid absorption, quick onset of action, and short duration of effect. These medications are taken orally before or during meals and can reduce HbA1c by 0.5%–1.5%.
Indications:
- Similar to SUs, glinides are particularly suitable for early-stage T2DM patients with postprandial hyperglycemia or elderly patients primarily experiencing postprandial hyperglycemia.
Contraindications or Special Considerations:
Adverse Reactions:
- Glinides may cause hypoglycemia and weight gain, though these effects are less pronounced than with SUs.
Clinical Application:
- Repaglinide: A benzoic acid derivative, with a dosage of 0.5–4 mg taken three times daily.
- Nateglinide: A D-phenylalanine derivative, with a dosage of 60–120 mg taken three times daily.
- Mitiglinide Calcium: Dosage of 10 mg taken three times daily.
Peroxisome Proliferator-Activated Receptor (PPAR) Activators
Thiazolidinediones (TZDs)
These medications activate PPARγ, increasing insulin sensitivity in target tissues. They also promote the redistribution of fat from visceral stores to subcutaneous stores. TZDs can reduce HbA1c by 0.7%–1.0%.
Indications:
- Used as monotherapy or in combination with other glucose-lowering medications for T2DM, especially in obese patients or those with significant insulin resistance.
Contraindications or Special Considerations:
- Not suitable for patients with T1DM, pregnant or lactating women, or children. Contraindicated in patients with heart failure (New York Heart Association [NYHA] Class II or higher), active liver disease, or liver transaminase levels exceeding 2.5 times the upper normal limit. Contraindicated in patients with a history of severe osteoporosis or bone fractures, and those with current or prior bladder cancer or unexplained visible hematuria.
Adverse Reactions:
- Monotherapy does not typically cause hypoglycemia, but the risk increases when combined with insulin or other insulin secretagogues. Common side effects include weight gain and edema, which are more pronounced when combined with insulin. Additional risks include fractures and heart failure.
Clinical Application:
- Rosiglitazone: 4–8 mg/day, taken once daily or in two divided doses.
- Pioglitazone: 15–30 mg/day, taken once daily.
Chiglitazar Sodium
Chiglitazar sodium moderately activates PPARα, PPARγ, and PPARδ, inducing expression of target genes associated with improved insulin sensitivity, fatty acid oxidation, energy metabolism, and lipid transport. It also inhibits PPARγ phosphorylation, which is linked to insulin resistance. Chiglitazar sodium can reduce HbA1c by 1.4%.
Indications:
- Used as monotherapy or in combination with other glucose-lowering medications for T2DM, particularly in obese patients, those with significant insulin resistance, or those with lipid metabolism disorders.
Contraindications or Special Considerations:
- Not recommended for patients with T1DM or diabetic ketoacidosis (DKA).
Adverse Reactions:
- Typically does not cause hypoglycemia when used alone, but the risk increases when combined with insulin or insulin secretagogues. May cause edema and weight gain, though these effects are relatively mild.
Clinical Application:
- Dosage: 32–48 mg, taken once daily.
Alpha-Glucosidase Inhibitors (AGI)
Alpha-glucosidase inhibitors work by inhibiting alpha-glucosidase enzymes at the brush border of the small intestinal mucosa, delaying carbohydrate absorption and reducing postprandial hyperglycemia. These medications can lower HbA1c by 0.5%–0.8% and do not cause weight gain.
Indications:
- Alpha-glucosidase inhibitors are suitable for individuals whose primary carbohydrate intake comes from sugar-based foods and who experience significant postprandial hyperglycemia while having normal or slightly elevated fasting glucose levels. These medications can be used as monotherapy or in combination with other glucose-lowering agents. For patients with T1DM, adding AGI to insulin therapy helps to reduce postprandial blood glucose levels and glucose variability.
Contraindications or Special Considerations:
- While AGIs generally do not cause systemic toxic reactions, caution is advised in patients with liver or kidney dysfunction. They are not suitable for those with gastrointestinal disorders, pregnant or breastfeeding women, and children. T1DM patients should not use AGIs as monotherapy.
Adverse Reactions:
- The most common adverse effects are gastrointestinal symptoms. Starting with a low dose and gradually increasing the dosage can minimize these effects. AGIs are not associated with hypoglycemia when used alone but may cause hypoglycemia when combined with sulfonylureas or insulin. In cases of hypoglycemia, oral or intravenous glucose is required, as double sugars or starchy foods cannot effectively alleviate the condition.
Clinical Application:
- Acarbose: 50–100 mg, three times daily.
- Voglibose: 0.2–0.3 mg, three times daily.
- Miglitol: 50–100 mg, three times daily.
- Sangzhi Total Alkaloids Tablets: 50–100 mg, three times daily. AGIs should be taken immediately after the first bite of food.
Dipeptidyl Peptidase-4 (DPP-4) Inhibitors
DPP-4 inhibitors reduce the inactivation of endogenous GLP-1 by inhibiting DPP-4 activity, thereby increasing GLP-1 levels. This enhances glucose-dependent insulin secretion and decreases postprandial glucagon secretion. DPP-4 inhibitors can lower HbA1c by 0.5%–1.0% without causing weight gain.
Indications:
- These medications are used as monotherapy or in combination with other oral glucose-lowering agents or insulin for the treatment of T2DM.
Contraindications or Special Considerations:
- Not suitable for pregnant women, children, patients with hypersensitivity to DPP-4 inhibitors, or those with T1DM or diabetic ketoacidosis (DKA).
Adverse Reactions:
- The overall incidence of adverse reactions is low. DPP-4 inhibitors, when used alone, do not increase the risk of hypoglycemia. Reported side effects include hypersensitivity reactions, elevated liver enzymes, upper respiratory infections, pancreatitis, and joint pain. Cardiovascular safety is generally good. Alogliptin and saxagliptin have not been associated with an increased risk of cardiovascular events but may pose a risk for heart failure-related hospitalization.
Clinical Application:
- Saxagliptin: 5 mg, once daily.
- Sitagliptin: 100 mg, once daily.
- Vildagliptin: 50 mg, once or twice daily.
- Linagliptin: 5 mg, once daily.
- Alogliptin: 25 mg, once daily. Dosage adjustments are necessary for patients with renal impairment except for linagliptin.
Sodium-Glucose Cotransporter-2 Inhibitors (SGLT-2i)
SGLT-2 inhibitors reduce glucose reabsorption in the proximal renal tubule by inhibiting SGLT-2 activity in the luminal membrane. This lowers the renal glucose threshold, promotes urinary glucose excretion, and decreases blood glucose levels. SGLT-2 inhibitors can reduce HbA1c by 0.5%–1.2%, aid in weight loss, lower blood pressure, and reduce urinary protein excretion. They also lower uric acid and triglyceride (TG) levels while increasing HDL-C levels. Some SGLT-2 inhibitors reduce all-cause mortality and cardiovascular mortality in T2DM patients with atherosclerotic cardiovascular disease (ASCVD) and lower the risks of composite cardiovascular events, heart failure hospitalization, and composite renal events.
Indications:
- These medications are used as monotherapy or in combination with other glucose-lowering agents and insulin for T2DM.
Contraindications or Special Considerations:
- Not recommended for patients with T1DM or chronic malnutrition. Attention should be given to each formulation's eGFR requirements.
Adverse Reactions:
- The overall incidence of adverse reactions is low. SGLT-2 inhibitors, when used alone, do not increase the risk of hypoglycemia but may do so when combined with insulin or insulin secretagogues. Genital and urinary tract infections may occur, typically mild to moderate in severity. Some medications may increase the risks of amputation and fractures. Normoglycemic diabetic ketoacidosis (DKA) has been reported; patients require close monitoring, and the medication should be discontinued if DKA is diagnosed.
Clinical Application:
- Dapagliflozin: 5–10 mg, once daily.
- Canagliflozin: 100–300 mg, once daily.
- Empagliflozin: 10–25 mg, once daily.
- Ertugliflozin: 5 mg, once daily.
- Huanggliflozin: 5–10 mg, once daily.
Glucokinase Activators (GKA)
Glucokinase activators act on glucokinase targets in pancreatic islets, intestinal endocrine cells, and the liver, promoting glucose-stimulated insulin and GLP-1 secretion while reducing insulin resistance. GKAs can lower HbA1c by approximately 1%.
Indications:
- These medications are used as monotherapy or in combination with metformin for T2DM.
Contraindications or Special Considerations:
- Not suitable for patients with T1DM, DKA, or hyperglycemic hyperosmolar states.
Adverse Reactions:
- Potential adverse effects include elevated transaminase levels and dyslipidemia.
Clinical Application:
- Dorzagliatin: 75 mg, twice daily, administered within one hour before breakfast and dinner.
Injectable Preparations
Insulin
Insulin is an important and effective method for managing hyperglycemia.
Indications:
- T1DM.
- Severe acute or chronic complications of diabetes.
- Surgery, pregnancy, and childbirth.
- Newly diagnosed, thin diabetes patients who are difficult to distinguish from T1DM.
- Newly diagnosed T2DM patients with significant hyperglycemia.
- Unexplained and significant weight loss during the course of diabetes.
- Marked decline in pancreatic β-cell function in T2DM.
- Certain specific forms of diabetes.
Classification of Insulin and Insulin Analogs:
- Based on their source and chemical structure, insulin can be classified into animal insulin, human insulin, and insulin analogs. Based on onset and duration of action, insulin is categorized into short-acting, intermediate-acting, long-acting, and premixed insulin. Insulin analogs are grouped into rapid-acting, long-acting, premixed insulin analogs, and biphasic insulin analogs. Weekly insulin preparations that require only one injection per week have been approved, and inhaled insulin and "smart insulin" are under development.
Short-acting insulin has a rapid onset and short duration when administered subcutaneously and can also be used intravenously for emergency management of diabetic ketoacidosis (DKA). Both short-acting insulin and rapid-acting insulin analogs, when injected subcutaneously before meals, are primarily used to control postprandial hyperglycemia. Intermediate-acting insulin, such as neutral protamine Hagedorn (NPH) insulin, is mainly used to provide basal insulin and can control postprandial hyperglycemia between meals. Long-acting insulins, such as protamine zinc recombinant human insulin (PZI) and long-acting insulin analogs, provide basal insulin without a pronounced action peak.
Table 4 Key characteristics of insulin and insulin analogue preparations (subcutaneous injection)
Insulin analogs are synthesized using DNA recombinant technology with modifications to their amino acid sequences. These analogs bind to the insulin receptor and exhibit similar functionality and effects as human insulin. Multiple insulin analogs are available on the market, including rapid-acting, long-acting, premixed preparations, and biphasic insulin analogs, to meet diverse clinical needs. While insulin analogs are similar to human insulin in terms of glycemic control, they offer advantages in mimicking physiological insulin secretion and reducing the risk of hypoglycemia.
Common rapid-acting insulin analogs:
- Insulin lispro.
- Insulin aspart.
- Insulin glulisine.
Modifications in the amino acid sequences of these analogs reduce their self-aggregation tendency, allowing them to remain in monomeric or dimeric forms. These modifications accelerate absorption after subcutaneous injection, with an onset of action typically observed within 15 minutes, reaching peak action in 30–60 minutes and lasting for 2–5 hours. This profile better aligns with physiological needs around meals, and these analogs are injected before meals.
Common long-acting insulin analogs:
- Insulin glargine.
- Insulin detemir.
- Insulin degludec.
Long-acting insulin analogs provide stable basal insulin levels, enabling better blood glucose control and a reduced risk of symptomatic and nocturnal hypoglycemia.
The type of insulin preparation, injection technique, injection site, variability in individual patient response, and the formation of insulin antibodies can all influence the onset of action, intensity, and duration of insulin's effects. Insulin should not be stored in freezing conditions, or exposed to excessive heat, extreme temperatures, or vigorous shaking. Premixed insulin preparations have fixed ratios and are only appropriate for patients with well-controlled and stable glucose levels.
Insulin "pen" injectors contain pre-filled insulin (or insulin analog cartridges), offering convenience and portability. Patients initiating insulin therapy require education to understand proper injection techniques. Ongoing follow-up is necessary after beginning insulin therapy, with an emphasis on encouraging and guiding patients to engage in self-monitoring of blood glucose levels.
Principles and Methods of Use
Insulin therapy should be implemented as part of a comprehensive treatment plan.
The treatment regimen should aim to mimic the physiological insulin secretion pattern.
Initiation of therapy should start with low doses, with gradual adjustments to achieve appropriate glucose control based on blood glucose levels.
Type 1 Diabetes Mellitus (T1DM):
Insulin therapy should be initiated immediately upon diagnosis and continued as lifelong replacement. Given the variability in residual β-cell quantity and function among patients, treatment regimens should be individualized.
Some patients with T1DM or those with slowly progressive immune-mediated diabetes in adults may initially use premixed insulin (or insulin analogs) with 2–3 injections per day. However, premixed insulin is not suitable for long-term therapy in T1DM.
Most patients require multiple daily subcutaneous insulin injections or continuous subcutaneous insulin infusion (CSII), particularly when β-cell function is severely impaired or during pregnancy. The initial dosage typically ranges from 0.2–0.4 U/(kg·d), with 40%–60% of the total daily dose provided as basal insulin and the remainder distributed among premeal doses. Short-acting insulin (administered subcutaneously 20–30 minutes before meals) or rapid-acting insulin analogs (administered immediately before meals) can be used for premeal injections. Intermediate- or long-acting insulin (or long-acting insulin analogs) is administered at bedtime. For patients with very poor β-cell function and significant glycemic variability, an additional small dose of intermediate- or long-acting insulin may be administered before breakfast to maintain daytime basal insulin levels.
CSII, which closely approximates physiological insulin secretion patterns, is considered a more optimal insulin therapy with a lower risk of hypoglycemia. Furthermore, innovations such as closed-loop insulin pump systems hold promise for the development of an "artificial pancreas," making this technology a bright prospect for T1DM management.
Type 2 Diabetes Mellitus (T2DM):
Insulin therapy should be considered under the following conditions:
- Persistent hyperglycemia despite lifestyle interventions and high-dose combinations of multiple oral hypoglycemic agents.
- Unexplained and significant weight loss during the course of the disease.
- Newly diagnosed T2DM with significant hyperglycemia or prominent symptoms. In such cases, insulin therapy can begin at diagnosis, with or without concomitant use of other glucose-lowering agents.
Treatment options include basal insulin (e.g., continuing oral hypoglycemic therapy during the day with bedtime injections of intermediate-acting insulin or long-acting insulin analogs), premixed insulin, or biphasic insulin analogs. Based on the patient's blood glucose levels, injections may be administered once or twice daily. When adopting a twice-daily injection regimen, insulin secretagogues should be discontinued.
Indications for long-term insulin replacement therapy in T2DM include significant β-cell dysfunction, poor response to oral hypoglycemic agents accompanied by weight loss or persistent hyperglycemia, and diabetes that is difficult to classify in lean patients. Treatment options may involve:
- Two daily injections of premixed insulin or 2–3 daily injections of premixed insulin analogs.
- Multiple daily injections combining basal and pre-meal insulin ("basal-bolus" therapy).
- CSII.
A trial regimen may be developed for each patient, with gradual adjustments made until satisfactory blood glucose control is achieved.
Possible Causes of High Fasting Blood Glucose Levels Despite Insulin Therapy:
- Insufficient nighttime basal insulin dosage.
- Dawn phenomenon: This refers to a brief period of hyperglycemia in the early morning hours, despite good glycemic control overnight and no occurrences of hypoglycemia. This is often attributed to increased secretion of hormones such as cortisol and growth hormone in the early morning.
- Somogyi phenomenon: This occurs when nighttime hypoglycemia, unnoticed during sleep, triggers a rebound hyperglycemic response due to the release of counter-regulatory hormones. Frequent blood glucose measurements during the night can aid in differentiating the underlying causes of morning hyperglycemia.
While intensive insulin therapy improves glycemic control, it increases the risk of hypoglycemia, which requires early identification and management. Intensive insulin therapy may not be suitable for infants under 2 years of age, elderly patients, individuals with severe complications, or those at high risk of hypoglycemia.
Acute stress often exacerbates metabolic imbalances in diabetic patients. Regardless of diabetes type or prior treatment regimen, insulin therapy is essential to manage hyperglycemia during acute episodes. Short-acting insulin should be added to intravenous glucose infusions as needed. Once the acute stress resolves, the diabetes treatment plan should be re-evaluated. For critically ill patients, current guidelines recommend maintaining blood glucose levels within the range of 7.8–10.0 mmol/L.
Adverse Reactions
The primary adverse reaction of insulin therapy is hypoglycemia, which is associated with overdose, improper injection techniques, and/or dietary imbalance. During the initial stages of insulin therapy, mild edema may occur due to sodium retention and typically resolves on its own. Some patients may experience temporary blurred vision caused by refractive changes in the lens, which generally resolves within a few weeks.
Different insulin preparations exhibit antigenicity and allergenicity due to their source, structure, composition, and the presence of excipients or impurities. Insulin allergic reactions commonly present as itching or urticaria-like rashes at the injection site, while severe allergic reactions are rare. Management strategies include switching the insulin preparation, using antihistamines and corticosteroids, and implementing desensitization therapy. In severe cases, discontinuation or temporary suspension of insulin therapy may be necessary.
Lipoatrophy or lipohypertrophy can occur at injection sites, involving subcutaneous fat loss or proliferation. Fatty tissue may gradually recover after injections are discontinued at the affected site. Regular rotation of injection sites helps to prevent these issues.
GLP-1 Receptor Agonists (GLP-1RAs)
GLP-1RAs stimulate insulin synthesis and secretion in a glucose-dependent manner, reduce glucagon secretion, and regulate appetite through action on the central nervous system. They also promote thermogenesis in brown adipose tissue, facilitate lipolysis in white adipose tissue, increase energy expenditure, and delay gastric emptying.
Currently available GLP-1RAs are administered via subcutaneous injection. These agents reduce HbA1c levels by approximately 1.0%–1.5% and are effective in promoting significant weight loss. Available or soon-to-be-available formulations include:
- Daily Injectable Formulations: Exenatide, lixisenatide, liraglutide, albiglutide, benaglutide, and semaglutide.
- Weekly Injectable Formulations: Semaglutide, dulaglutide, long-acting exenatide, and efpeglenatide.
- Oral semaglutide tablets have also been launched. Additionally, tirzepatide, a GLP-1/GIP dual receptor agonist, has been approved for T2DM management, and GIP/GLP-1/glucagon triple receptor agonists are under development.
Indications:
- GLP-1RAs are approved for use in the treatment of T2DM, either as monotherapy or in combination with other glucose-lowering agents, particularly in patients with obesity or significant insulin resistance. Certain GLP-1RAs have demonstrated cardiovascular protective effects, making them suitable for T2DM patients with atherosclerotic cardiovascular disease (ASCVD) or those at high cardiovascular risk.
Contraindications or Precautions:
- GLP-1RAs are contraindicated in patients with T1DM, diabetic ketoacidosis (DKA), type 2 multiple endocrine neoplasia, or a history or family history of medullary thyroid carcinoma. These agents should be used cautiously in patients with a history of pancreatitis, gallbladder disease, gastroparesis, or prior gastric surgery. Renal function requirements for each formulation must be considered, and semaglutide should be used with caution in patients with proliferative diabetic retinopathy.
Adverse Reactions:
- Common adverse reactions include gastrointestinal discomfort such as nausea, vomiting, diarrhea, appetite suppression, and abdominal bloating. These symptoms tend to diminish with continued treatment. Other reported reactions include upper respiratory tract infections and injection site nodules. The incidence of hypoglycemia is low. GLP-1RAs may cause an increase in heart rate, which can lead to palpitations. Rare adverse events include pancreatitis and dermatitis. Clinical studies have reported that semaglutide may exacerbate diabetic retinopathy in some patients during the early stages of treatment.
Clinical Applications:
- Exenatide: Initial dose of 5 μg, administered twice daily, with an increase to 10 μg twice daily as needed. Long-acting exenatide is administered once weekly.
- Liraglutide: Initial dose of 0.6 mg once daily, with the dosage gradually increased to 1.2–1.8 mg once daily.
- Benaglutide: Initial dose of 0.1 mg three times daily.
- Semaglutide: Initial dose of 0.25 mg once weekly; the maximum recommended dose for diabetes treatment is 1 mg per week.
- Dulaglutide: Initial dose of 0.75 mg once weekly; the maximum recommended dose is 1.5 mg per week.
- Efpeglenatide: Initial dose of 0.1 mg once weekly; the maximum recommended dose is 0.2 mg per week.
Weight Management and Metabolic Surgery
Weight management is a crucial aspect of comprehensive diabetes care. For overweight or obese patients, weight loss helps improve glycemic control, reduces the need for glucose-lowering medications, and contributes to managing ASCVD risk factors. The first approach involves lifestyle interventions, with adjunctive use of weight-loss medications when necessary. The impact of medications on weight should be considered when selecting glucose-lowering drugs. Medications such as SGLT-2 inhibitors, GLP-1 receptor agonists, and GLP-1/GIP dual receptor agonists show promising potential for both glycemic control and weight reduction in diabetes treatment.
For patients who fail to achieve effective weight loss or adequate glycemic control despite lifestyle interventions with or without pharmacotherapy, metabolic surgery may be considered. Studies have demonstrated that metabolic surgery significantly improves weight, hyperglycemia, and dyslipidemia in patients with obesity and T2DM. The procedure should be conducted in large medical centers with multidisciplinary teams experienced in managing diabetes and gastrointestinal surgical diseases. A comprehensive preoperative evaluation is needed to assess patient adherence, psychological health, history of alcohol or drug abuse, and any relevant psychiatric disorders. Post-surgery, patients should receive long-term lifestyle support and lifelong follow-up care.
Currently, there is no complete consensus on the indications, contraindications, and specific procedures for metabolic surgery. Most existing clinical evidence is based on non-Asian populations. It is important to strictly regulate the eligibility criteria for surgery, weigh the risks and benefits, and ensure effectiveness while minimizing the risks of short- and long-term complications.
Pancreas Transplantation and Islet Cell Transplantation
A successful pancreas transplantation or combined pancreas-kidney transplantation can eliminate the need for exogenous insulin administration and improve quality of life. The main candidates for such procedures are T1DM patients, particularly those with end-stage renal disease, or those who, despite intensive insulin therapy, fail to achieve adequate glycemic control and experience recurrent severe metabolic dysregulation. However, long-term use of immunosuppressive drugs is required following transplantation.
Allogeneic islet cell transplantation allows some T1DM patients to maintain near-normal blood glucose levels for several years. However, the scarcity of donors and the requirement for lifelong immunosuppression limit its clinical application. Additionally, the percentage of patients who remain insulin-independent five years after transplantation is less than 10%. Recent advances in cell-based therapies for T1DM, particularly stem cell therapies, show promising potential, but these remain in the preclinical research stage.
Principles for Preventing and Managing Chronic Diabetes Complications
Chronic complications of diabetes are the leading causes of disability and death in patients, highlighting the importance of early prevention and intervention. For T1DM patients with a disease duration of five years or more, and for all T2DM patients beginning at diagnosis, annual screening for chronic complications is recommended. Early, comprehensive, and aggressive control of ASCVD risk factors is vital.
Blood Pressure Management
Blood pressure monitoring at home is recommended for all patients with concomitant hypertension. Target blood pressure is generally below 130/80 mmHg. Medication options include angiotensin-converting enzyme inhibitors (ACEIs), angiotensin II receptor blockers (ARBs), calcium channel blockers (CCBs), low-dose diuretics, selective beta-blockers, and angiotensin receptor-neprilysin inhibitors. ACEIs or ARBs are preferred, often in combination with other antihypertensive agents. For patients whose blood pressure remains uncontrolled despite the use of three types of antihypertensive medications (including a diuretic), mineralocorticoid receptor antagonists (MRAs) may be considered.
Lipid Management:
Lifestyle interventions form the foundation of lipid management in diabetes, with achieving target LDL-C levels being the primary objective. A comprehensive assessment of ASCVD risk should guide the treatment of dyslipidemia. The guidelines recommend the following LDL-C targets:
- LDL-C <1.4 mmol/L for T2DM patients with ASCVD.
- LDL-C <1.8 mmol/L for T2DM patients at high ASCVD risk.
- LDL-C <2.6 mmol/L for T2DM patients at low-to-moderate ASCVD risk.
Statins are the first-line agents and should be used long-term. For patients with triglycerides (TG) >5.6 mmol/L, fibrates, high-purity omega-3 fatty acids, or niacin are recommended to reduce the risk of acute pancreatitis. For patients intolerant to statins, alternatives like cholesterol absorption inhibitors or PCSK9 inhibitors may be considered.
For patients who fail to achieve LDL-C targets with statins or who have uncontrolled mixed dyslipidemia, combination therapy can be used, such as statins with cholesterol absorption inhibitors, or statins with fibrates or omega-3 fatty acids. For patients at extremely high cardiovascular risk with high baseline LDL-C levels, or for those unlikely to meet LDL-C targets even with statin combination therapies, PCSK9 inhibitors may be added to further reduce the risk of cardiovascular events.
Regular follow-up to monitor treatment efficacy and adverse effects, along with adjustments to therapy, is essential to maintain long-term lipid control.
Use of Low-Dose Aspirin
Low-dose aspirin (75–150 mg/day) is recommended for secondary prevention in diabetes patients with a history of ASCVD. For those who cannot tolerate aspirin, clopidogrel (75 mg/day) can be used as an alternative. For patients aged ≥50 years who have ASCVD risk factors but no increased risk of bleeding, low-dose aspirin may be considered as a primary prevention strategy.
Prevention and Management of Diabetic Kidney Disease (DKD)
Strict glycemic control is important in preventing or delaying the onset and progression of DKD. For T2DM patients with high cardiovascular and renal risks, glucose-lowering drugs with renal benefits — such as SGLT-2 inhibitors and GLP-1 receptor agonists — should be prioritized. In early kidney disease characterized by microalbuminuria with normal blood pressure, the use of ACEIs or ARBs has been shown to slow the progression of kidney disease. Once clinical-stage kidney disease develops, treatment should focus on controlling hypertension and slowing the decline of GFR.
In such cases, ACEIs or ARBs not only lower blood pressure but also reduce proteinuria and delay GFR decline. Additionally, the combination with novel nonsteroidal mineralocorticoid receptor antagonists can further improve blood pressure control, reduce proteinuria, and lower the risks of both renal and cardiovascular events. For patients with UACR ≥300 mg/g, a reduction of UACR by 30% or more is recommended to slow disease progression. Dietary protein intake should be moderately restricted, with a preference for high-quality animal protein. Supplements such as compound α-keto acids may further help to slow GFR decline. Early intervention with erythropoiesis-stimulating agents or hypoxia-inducible factor prolyl hydroxylase inhibitors (HIF-PHI) can help correct anemia. Correcting vitamin D, calcium, and phosphorus imbalances significantly improves the quality of life and prognosis for advanced-stage patients. Renal replacement therapy should be initiated earlier than in non-diabetic kidney disease patients.
Comprehensive Management of Diabetic Retinopathy
Comprehensive ophthalmic evaluations should include pupil-dilated fundoscopy and color fundus photography, with fluorescein fundus angiography performed if necessary. Optical coherence tomography (OCT) provides a clearer depiction of retinal vascular spatial distribution and enables the early detection of retinal vascular abnormalities. Its non-invasive nature also makes it suitable for follow-up assessments.
Patients with severe nonproliferative diabetic retinopathy (NPDR) are recommended to undergo panretinal laser photocoagulation promptly. In cases of proliferative diabetic retinopathy (PDR) where vision is threatened (e.g., persistent vitreous hemorrhage, or complications such as cataracts, iris neovascularization, or fibrous proliferation causing retinal traction or detachment), vitrectomy should be performed promptly. Anti-vascular endothelial growth factor (VEGF) drugs have become the first-line treatment for vision-threatening diabetic macular edema. For those with access, subthreshold micropulse laser combined with anti-VEGF therapy is recommended. Close monitoring is especially important during pregnancy.
Management of Diabetic Neuropathy
Early and prolonged stable glycemic control is the most critical and effective strategy for preventing and managing diabetic neuropathy. Pharmacological therapies such as mecobalamin, prostaglandin analogs, aldose reductase inhibitors, and α-lipoic acid have some efficacy. For severe neuropathic pain, symptomatic treatments may include anticonvulsants, selective serotonin and norepinephrine reuptake inhibitors (SNRIs), tricyclic antidepressants, or opioid drugs.
Comprehensive Foot Care
All patients should regularly undergo foot evaluations, including physical examinations, testing protective sensation, and assessing for peripheral arterial disease. Education on self-care of the feet is essential. In high-risk feet, trauma should be avoided, and vascular and neurological conditions should be managed proactively. For patients with foot ulcers or high-risk feet, multidisciplinary care and standardized treatment are recommended to reduce amputation rates and healthcare costs.
Management of Hyperglycemia During Pregnancy
Hyperglycemia during pregnancy is associated with maternal and neonatal complications, including spontaneous abortion, preeclampsia, fetal malformations or death, macrosomia, neonatal hypoglycemia, hyperbilirubinemia, and respiratory distress syndrome. Good glycemic control both before and during pregnancy reduces these risks. Diabetic women should plan their pregnancies, with pre-pregnancy evaluations performed jointly by diabetes and obstetrics specialists to assess suitability for pregnancy.
Elevated HbA1c levels before the 10th week of pregnancy correlate with increased risk of maternal and neonatal complications. Female diabetes patients should achieve optimal glycemic control (ideally HbA1c <6.5%) through insulin therapy before conceiving. Gestational diabetes mellitus (GDM) should be diagnosed as early as possible, and management should adhere to standard clinical guidelines once diagnosed.
The goals of medical nutrition therapy during pregnancy include meeting the nutritional needs of both the mother and fetus, maintaining normal blood glucose levels, and avoiding starvation ketosis. Many GDM patients can achieve glycemic targets through lifestyle interventions alone. If additional therapy is required, insulin remains the treatment of choice, administered via multiple daily injections or continuous subcutaneous insulin infusion (CSII). At present, no other glucose-lowering medications have been approved for gestational hyperglycemia. Continuous monitoring of blood glucose is essential, and time-in-range (TIR) metrics can assist in achieving glycemic goals during pregnancy for T1DM patients. Recommended glycemic targets during pregnancy are as follows: fasting plasma glucose (FPG) 3.9–5.3 mmol/L, postprandial 1-hour plasma glucose (1hPG) <7.8 mmol/L, postprandial 2-hour plasma glucose (2hPG) <6.7 mmol/L, and HbA1c <6.0%, provided that significant hypoglycemia does not occur.
Close monitoring of fetal development, maternal blood pressure, renal function, and fundus is recommended. Delivery timing and mode should be determined based on the condition of both the fetus and the mother.
Postpartum, attention should be given to preventing and managing neonatal hypoglycemia. GDM patients are encouraged to breastfeed to reduce the risk of developing T2DM postpartum. Screening for persistent diabetes is recommended 4–12 weeks after delivery. If postpartum blood glucose is normal, diabetes screening should be conducted at least every three years.
Perioperative Management
Before elective surgery, a comprehensive evaluation of diabetes-related complications is recommended, with the aim of reducing HbA1c levels to less than 8%. For patients undergoing major or medium-sized surgical procedures, insulin therapy should be initiated or switched to at least three days prior to the surgery. A basal-bolus multiple daily injection insulin regimen or continuous subcutaneous insulin infusion (CSII) is generally preferred. In cases requiring emergency surgery, where acid-base or fluid-electrolyte imbalances are present, these imbalances should be corrected promptly. Close blood glucose monitoring is crucial, and maintaining perioperative blood glucose levels between 7.8 and 10.0 mmol/L is considered relatively safe. For patients without significant hypoglycemia, stricter targets may be appropriate, particularly for those undergoing cardiac or other precision surgery, where glucose levels may be maintained at 6.1–7.8 mmol/L or 5.6–10.0 mmol/L. Postoperative recovery involves adjustment of diabetes treatment regimens based on the patient's condition.
Post-Transplant Diabetes
The use of immunosuppressive agents following organ transplantation may result in glucose metabolism abnormalities, which can negatively affect transplanted organs. Screening and management of hyperglycemia are essential in these cases.
Immunization
Vaccination according to age for both children and adult diabetes patients with routinely recommended vaccines is advised.
Prevention
Efforts to prevent, treat, educate, and manage diabetes require collaboration from governments, health authorities, and various sectors of society. Promoting healthy lifestyles and enhancing the management of high-risk populations is essential. Overweight and obese individuals are encouraged to achieve and maintain a healthy body weight. Medications such as metformin, alpha-glucosidase inhibitors, and pioglitazone can be used to prevent or delay the onset of T2DM in high-risk individuals. Teplizumab has been approved by the FDA for the prevention of T1DM.