Hypoglycemia is a clinical syndrome characterized by a reduction in plasma (or serum) glucose levels leading to corresponding symptoms and signs. Symptoms primarily manifest as autonomic nervous system activation and/or neuropsychiatric and behavioral abnormalities. At lower blood glucose concentrations, seizures, coma, and even death may occur. Symptoms and signs resolve as plasma glucose concentration increases. The plasma glucose threshold for triggering hypoglycemic symptoms typically ranges from 2.8 to 3.9 mmol/L, although this threshold may be lower in patients with recurrent episodes of hypoglycemia.
Hypoglycemia can occur in both diabetic and non-diabetic individuals. In diabetic patients, hypoglycemia is often related to glucose-lowering treatments, with the primary goal being the adjustment of therapeutic regimens to minimize or prevent its occurrence. In non-diabetic individuals, accurate identification of the underlying cause is required to select an appropriate treatment strategy. Hypoglycemia can be broadly categorized into insulin-mediated and non-insulin-mediated subtypes based on its underlying mechanisms.
Etiology
Hypoglycemia in Non-Diabetic Individuals
Drugs
Although relatively uncommon, certain non-antidiabetic medications can induce hypoglycemia. These include ethanol, quinolones, pentamidine, quinine, beta-blockers, angiotensin-converting enzyme inhibitors (ACEIs), and IGF-1, among others.
Diseases Associated with Hypoglycemia
Hypoglycemia-related diseases can be divided into non-insulin-mediated and insulin-mediated categories:
Non-Insulin-Mediated Hypoglycemia
This is often caused by severe illnesses, including liver failure, kidney failure, heart failure, sepsis, or malnutrition.
Non-pancreatic cell tumors can also lead to hypoglycemia, commonly occurring in large mesenchymal or epithelial tumors. Hypoglycemia in these cases typically results from tumor production of incompletely processed insulin-like growth factor 2 (IGF-2), which inhibits endogenous insulin synthesis.
Hormonal deficiencies affecting insulin counter-regulation may contribute, including adrenal insufficiency, growth hormone deficiency, glucagon deficiency, or myxedema coma. Plasma insulin levels in non-insulin-mediated hypoglycemia are low or within the normal range.
Insulin-Mediated Hypoglycemia
Also referred to as endogenous hyperinsulinemia, this occurs when plasma glucose levels drop to hypoglycemic levels without a corresponding decrease in insulin secretion. In non-diabetic adults, this can result from several conditions:
- Insulinoma: Tumors of pancreatic beta cells that secrete insulin inappropriately.
- Functional Disorders of Pancreatic Beta Cells: Non-insulinoma pancreatogenous hypoglycemia syndrome (NIPHS) is an example. Post-gastric bypass surgery, food may rapidly enter the intestine, leading to postprandial hyperglycemia, increased glucagon-like peptide secretion, and excessive insulin release. This suppresses hepatic glucose production while enhancing glucose utilization, resulting in relative postprandial hyperinsulinemia and subsequent hypoglycemia.
- Insulin Autoimmune Hypoglycemia: This occurs in patients with antibodies against endogenous insulin or insulin receptors. Symptoms of hypoglycemia may appear either postprandially, during fasting, or both. Insulin forms a complex with antibodies and later dissociates in an unregulated manner, leading to hyperinsulinemia and hypoglycemia. In patients with insulin receptor antibodies, hypoglycemia is caused by stimulatory antibodies activating insulin receptors.
- Drug-Induced Hyperinsulinemia in Non-Diabetic Individuals: Endogenous hyperinsulinemia may also occur due to the use of insulin secretagogues, even in non-diabetic patients. In cases of accidental, concealed, or unclear causes of hypoglycemia, the possibility of medication errors, drug-related factors, or patient-related misuse of insulin secretagogues should be considered. This includes accidental ingestion of medications prescribed for diabetic household members or the covert use of antidiabetic drugs or insulin by some individuals.
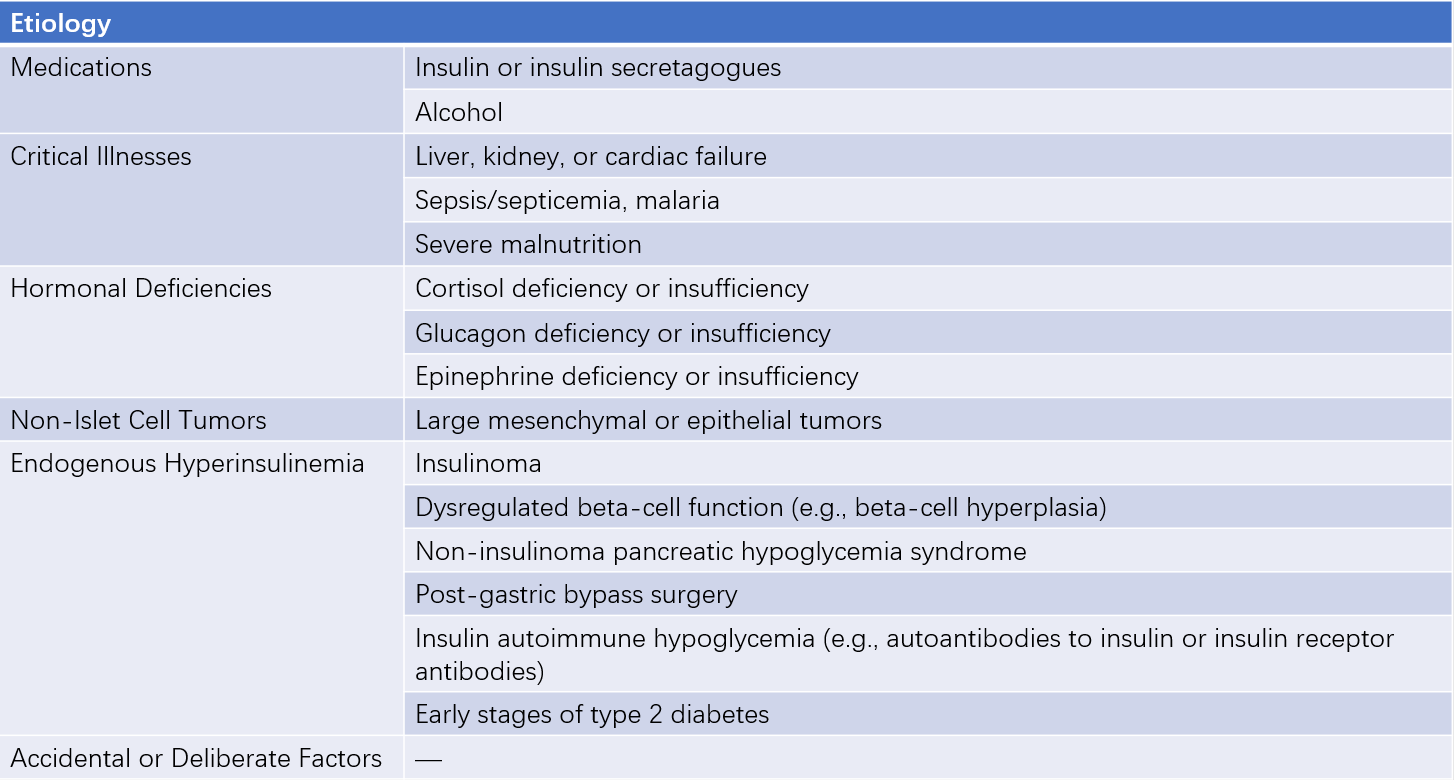
Table 1 Causes of hypoglycemia
Persistent hyperinsulinemic hypoglycemia of infancy (PHHI) or congenital hyperinsulinemia is the most common cause of persistent hypoglycemia in infants.
Hypoglycemia in Diabetic Patients
In the early stages of type 2 diabetes, delayed insulin release can lead to discordance between postprandial insulin levels and blood glucose levels, resulting in hypoglycemia. Exogenous insulin and medications that stimulate endogenous insulin secretion (such as insulin secretagogues like glibenclamide, gliclazide, glipizide, glimepiride, repaglinide, and nateglinide) can promote glucose utilization, potentially leading to hypoglycemia if misused. Severe or even fatal hypoglycemia may occur in such cases. Oral antidiabetic drugs such as insulin sensitizers, alpha-glucosidase inhibitors, glucagon-like peptide-1 (GLP-1) receptor agonists, sodium-glucose co-transporter-2 (SGLT-2) inhibitors, and dipeptidyl peptidase-4 (DPP-4) inhibitors carry a much lower risk of causing hypoglycemia. These drugs exert their effects primarily through mechanisms such as enhancing insulin sensitivity, inhibiting monosaccharide absorption, or promoting glucose excretion through the urine. As plasma glucose concentrations return to the normal range, insulin secretion appropriately decreases.
Although GLP-1 receptor agonists can stimulate insulin secretion, they do so in a largely glucose-dependent manner. They also suppress glucagon secretion in a glucose-dependent fashion. When glucose levels fall below a certain threshold, insulin secretion decreases, and glucagon secretion increases, resulting in a lower risk of hypoglycemia. Nonetheless, when combined with insulin secretagogues or exogenous insulin, all of these medications have the potential to increase the risk of hypoglycemia.
Pathophysiology
The brain relies almost exclusively on glucose as its energy source. Because the brain cannot synthesize or store glucose, it requires a continuous supply of glucose from circulation to maintain normal functioning and physiological needs. Under normal physiological conditions, fasting plasma glucose is maintained within a range of 3.9–6.0 mmol/L. When blood glucose levels fall below the physiological range and are insufficient to meet the brain's energy demands, the body utilizes finely tuned glucose-raising mechanisms to maintain blood glucose within the normal range.
Blood glucose homeostasis depends on a complex regulatory network involving neural signals, hormones, and metabolic substrates, with insulin playing a central role. As plasma glucose levels decline, insulin secretion decreases, triggering glycogenolysis and gluconeogenesis to restore blood glucose to physiological levels. The suppression of insulin secretion acts as the first line of defense against hypoglycemia under normal conditions.
When blood glucose drops below the physiological range, the secretion of glucose-raising hormones increases. Elevated glucagon levels, produced by alpha cells in the pancreas, serve as the second line of defense against hypoglycemia. If glucagon secretion is insufficient to correct hypoglycemia, epinephrine secretion increases, representing the third line of defense.
If hypoglycemia persists beyond four hours, cortisol and growth hormone secretion increases, promoting glucose production and limiting glucose utilization. However, the roles of glucocorticoids and growth hormone in defending against acute hypoglycemia are relatively minor. If these defense mechanisms fail to effectively restore blood glucose levels, further reductions in glucose concentration lead to the onset of hypoglycemic symptoms and signs.
The threshold for hypoglycemic symptoms and signs is not fixed and varies depending on the underlying cause, the frequency and duration of hypoglycemia, and individual patient factors. For example, poorly controlled diabetic patients often have a higher hypoglycemic threshold, and symptoms of hypoglycemia may occur within the normal glucose range (referred to as pseudohypoglycemia). Conversely, the threshold for hypoglycemia may be lower in certain conditions, such as in patients with recurrent hypoglycemia (e.g., those undergoing intensive glucose-lowering therapy or patients with insulinomas), where symptoms often present at much lower blood glucose levels.
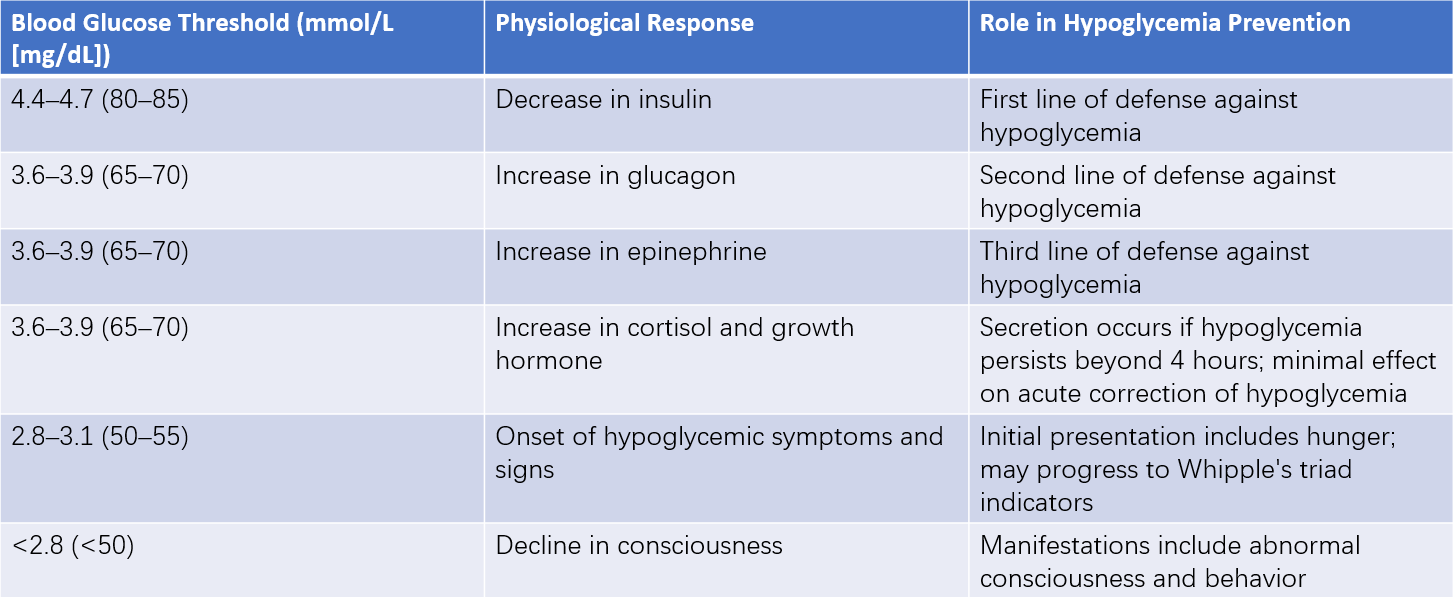
Table 2 Physiological responses to hypoglycemia and corresponding blood glucose thresholds
Clinical Manifestations
Symptoms
Before determining the cause of hypoglycemia through diagnostic tests, establishing evidence of hypoglycemia is critical. The presence of Whipple's triad is helpful for confirming hypoglycemia and its associated conditions. Whipple's triad consists of:
- Symptoms consistent with hypoglycemia.
- Measured low blood glucose concentration during the symptomatic episode, confirmed by precise methods (typically venous blood glucose rather than home glucometers).
- Resolution of symptoms once glucose levels are restored to normal.
Hypoglycemic symptoms are broadly classified into two categories: autonomic symptoms and neuroglycopenic symptoms.
Autonomic Symptoms
These include tremor, palpitations, and anxiety, which are catecholamine-mediated adrenergic symptoms, as well as sweating, hunger, and paresthesia, which are cholinergic symptoms mediated by acetylcholine. These symptoms largely stem from sympathetic nervous system activation rather than adrenal medullary activation.
Neuroglycopenic Symptoms
These include cognitive impairment, behavioral changes, psychomotor abnormalities, as well as seizures and coma that occur at lower blood glucose concentrations.
Signs
Pallor and perspiration are common signs of hypoglycemia. Heart rate and systolic blood pressure may increase, but the extent of these elevations is usually modest. Transient neurological deficits can occasionally occur. Permanent neurological damage can be seen in patients with prolonged, recurrent episodes of severe hypoglycemia or those who experience a single episode of severe hypoglycemia that is not promptly corrected.
Laboratory Testing
The initial goal of laboratory evaluation is to confirm Whipple's triad. If Whipple's triad has already been confirmed, the objective shifts to assessing whether hypoglycemia is insulin-mediated. For diabetic patients experiencing suspected hypoglycemia, blood glucose measurement is necessary, along with a comprehensive review of the current treatment regimen, types and doses of medications, timing in relation to meals, and physical activity levels. For non-diabetic individuals exhibiting symptoms suggestive of hypoglycemia, the first step is to confirm the presence of hypoglycemia, followed by obtaining information on blood glucose, insulin, related hormones, and metabolic markers to provide reliable clues for diagnosis and differential diagnosis. Laboratory evaluations for suspected hypoglycemia in non-diabetic patients should include the following:
Blood Glucose
The lower limit of normal fasting blood glucose is generally 3.9 mmol/L (70 mg/dL). In non-diabetic individuals, as blood glucose levels decrease within the physiological range, insulin secretion also decreases correspondingly. When blood glucose drops to 3.6–3.9 mmol/L (65–70 mg/dL), glucoregulatory hormones (glucagon and epinephrine) start to increase. These hormonal responses occur before hypoglycemic symptoms develop, which often appear at glucose levels of 2.8–3.1 mmol/L (50–55 mg/dL). It is important to note that the hypoglycemia threshold can vary, and clinical judgment should account for individual patient factors.
Measurement of Plasma-Related Hormones
Identifying the cause of hypoglycemia requires simultaneous measurement of blood glucose, insulin, C-peptide, beta-hydroxybutyrate, and insulin autoantibodies during a symptomatic episode. In addition, the blood glucose response to an injection of 1.0 mg of glucagon should be evaluated. These steps help distinguish between endogenous and exogenous insulin-mediated hypoglycemia and identify potential underlying causes.
Plasma (or serum) insulin levels should be measured when blood glucose concentrations are below 3.1 mmol/L (55 mg/dL). A plasma insulin level of 18 pmol/L (3 µU/mL) or higher, as measured by immunochemiluminescent assay (ICMA), indicates excessive insulin and meets the criteria for endogenous hyperinsulinemia, such as that seen in insulinoma.
Measuring plasma C-peptide levels and proinsulin can further differentiate between endogenous and exogenous hyperinsulinemia. In patients with blood glucose concentrations below 3.1 mmol/L (55 mg/dL), a plasma C-peptide concentration of 0.6 ng/mL (0.2 nmol/L) or higher confirms endogenous hyperinsulinemia. Due to insulin's ketogenesis-inhibiting effects, plasma beta-hydroxybutyrate concentrations in insulinoma patients are typically lower than in healthy individuals. At the end of a fasting test, beta-hydroxybutyrate levels in all insulinoma patients are 2.7 mmol/L or lower, whereas levels rise in healthy subjects. A gradual increase in beta-hydroxybutyrate concentration after 18 hours of fasting suggests a negative fasting test.
For patients with borderline insulin and C-peptide levels during hypoglycemic episodes, the diagnosis can be confirmed by evaluating plasma beta-hydroxybutyrate concentration and assessing blood glucose response to glucagon administration.
Diagnosis and Differential Diagnosis
For hypoglycemia in diabetic patients, the diagnosis of diabetes-related hypoglycemia can generally be made through a detailed history of diabetes and the use of glucose-lowering medications. For hypoglycemia in non-diabetic patients, further confirmation and differentiation are required, as the outcomes and clinical management of such cases differ significantly from diabetes-related hypoglycemia.
In non-diabetic patients, the diagnosis of hypoglycemia needs to be established first. Hypoglycemia can be confirmed based on its typical presentation (Whipple's triad), which includes:
- Symptoms of hypoglycemia.
- Blood glucose levels below 2.8 mmol/L during episodes.
- Rapid resolution of symptoms following administration of glucose.
For patients with unclear or mild reductions in fasting blood glucose, or those not in an active symptomatic phase, repeated testing to detect possible fasting or postprandial hypoglycemia may be necessary. A 48- to 72-hour fasting test can also be performed if required.
Measurements of plasma or serum insulin, C-peptide, beta-hydroxybutyrate, and counter-regulatory hormones (e.g., cortisol, growth hormone), along with functional tests, can provide insights into the potential causes of hypoglycemia. Insulin antibodies should be tested in cases of endogenous hyperinsulinemia to determine if the hypoglycemia is autoimmune in nature.
For hypoglycemia mediated by endogenous insulin, differential diagnoses include insulinoma, pancreatic islet cell hyperplasia/hypertrophy, hypoglycemia induced by oral antidiabetic drugs, and insulin autoimmune syndrome. For all insulin-mediated hypoglycemia excluding antibody- or drug-induced cases, localization studies such as CT, MRI, or abdominal ultrasound should be performed. Additional testing, such as endoscopic ultrasound, calcium stimulation tests, or somatostatin receptor imaging using isotopic techniques, may be required if necessary.
For hypoglycemia not mediated by insulin, differential diagnoses include hypoglycemia due to non-antidiabetic medications, liver disease, kidney disease, cardiac disease, systemic illnesses, extrapancreatic tumors, growth hormone deficiency, cortisol deficiency, hypopituitarism, and myxedema coma. Combining liver and kidney function tests, evaluations of counter-regulatory hormones, and imaging studies such as CT or MRI can help identify the underlying cause.
Prevention
Clinicians can propose prevention strategies by identifying diagnostic clues to hypoglycemia, including a history of diabetes, the course of glucose-lowering treatment (particularly the use of insulin secretagogues, insulin dosages, dietary and exercise patterns, and hypoglycemia in relation to meals), the use of non-antidiabetic drugs, history of alcohol abuse, and any systemic diseases (e.g., tumors, wasting illnesses, malnutrition, gastrointestinal surgeries). Blood glucose levels should be monitored in individuals with unexplained symptoms of brain dysfunction. Repeated severe episodes of prolonged hypoglycemia can lead to irreversible brain damage, so early recognition and timely management are vital. Further localization diagnostics should be performed for suspected insulinoma, and tumor resection should be considered for patients with confirmed insulinoma.
Treatment
Treatment involves two primary goals: resolving symptoms of insufficient glucose supply to the nervous system and addressing the underlying causes of hypoglycemia. Mild to moderate hypoglycemia can be alleviated by consuming glucose-containing beverages, sugar water, candy, biscuits, bread, or steamed buns. For drug-related hypoglycemia, discontinuation of the causative medication is recommended. In severe cases or in patients suspected of hypoglycemic coma, blood glucose levels should be measured promptly. However, even in the absence of blood glucose measurements, immediate intravenous injection of 60–100 mL of 50% glucose solution should be administered, followed by intravenous infusion of 5%–10% glucose solution. If necessary, hydrocortisone (100 mg) and/or glucagon (0.5–1 mg) can be administered intramuscularly or intravenously. For unconscious patients, feeding must be avoided to prevent airway obstruction.
For patients using insulin or insulin secretagogues in combination with alpha-glucosidase inhibitors, pure glucose should be used to treat symptomatic hypoglycemia. Since alpha-glucosidase inhibitors slow the digestion of other carbohydrates, other forms of carbohydrates (e.g., starches, sucrose) are ineffective for promptly treating hypoglycemia induced by this combination therapy.