Dyslipidemia generally refers to elevated levels of serum cholesterol (CH), triglycerides (TG), and low-density lipoprotein cholesterol (LDL-C), accompanied by reduced levels of high-density lipoprotein cholesterol (HDL-C). Since lipids in plasma exist in the form of lipoproteins, dyslipidemia manifests as dyslipoproteinemia. Dyslipidemia can lead to atherosclerosis, resulting in cardiovascular and cerebrovascular diseases such as coronary heart disease and stroke. The effective prevention and treatment of dyslipidemia are of significant importance for reducing the prevalence of cardiovascular disease and improving quality of life.
Lipids, Apolipoproteins, and Lipoproteins
Blood lipids refer to the total amount of neutral fats (cholesterol and triglycerides) and lipoid substances (phospholipids, glycolipids, sterols, and steroids) in plasma. In humans, cholesterol primarily exists in the forms of free cholesterol and cholesterol esters, while triglycerides (TG) are formed when the three hydroxyl groups of a glycerol molecule are esterified with fatty acids. Blood lipids are insoluble in water and are transported and utilized by combining with apolipoproteins to form lipoproteins.
Apolipoproteins serve as carriers for lipid transport, regulate enzyme activity related to lipid metabolism, and mediate recognition and binding to cell membrane receptors. Over 20 types of apolipoproteins have been identified and are categorized into apo A, apo B, apo C, apo D, and apo E. Each type is further divided into subtypes based on differences in amino acid sequences, such as apo A1, A2, A4; apo B48, B100; apo C1, C2, C3; and apo E1, E3, etc. Apolipoproteins also include one with variable length, apo(a), which binds to LDL.
Plasma lipoproteins are spherical macromolecular complexes comprising apolipoproteins, cholesterol (CH), triglycerides (TG), phospholipids (PL), and other components. They are classified into six major categories: chylomicrons (CM), very low-density lipoproteins (VLDL), intermediate-density lipoproteins (IDL), low-density lipoproteins (LDL), high-density lipoproteins (HDL), and lipoprotein(a) [Lp(a)].
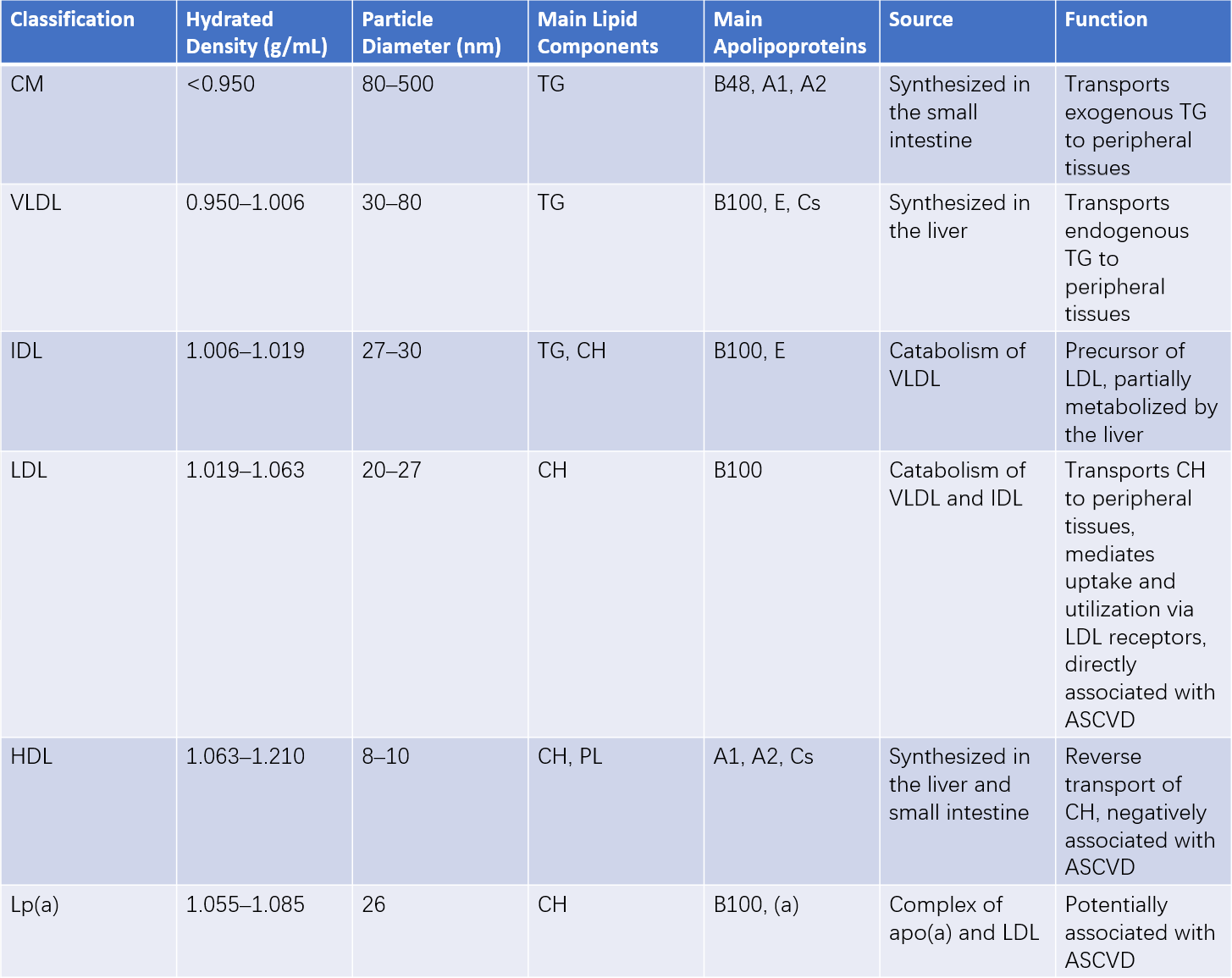
Table 1 Main characteristics and functions of lipoproteins
The metabolic pathways of lipoproteins include:
- An exogenous pathway, which involves the synthesis of CM from dietary CH and TG in the small intestine and their subsequent metabolism.
- An endogenous pathway, which entails the transformation of VLDL synthesized in the liver into IDL and LDL and the subsequent metabolism of LDL by the liver or other organs.
- A reverse cholesterol transport pathway, which involves HDL transporting CH from peripheral tissues to the liver for metabolism and recirculation.
Chylomicrons (CM)
These lipoproteins have the largest size and the lowest density, with TG accounting for approximately 90% of their content. Their primary role is to transport exogenous TG to extrahepatic tissues. CM particles do not penetrate arterial walls and generally do not cause atherosclerosis, but they are prone to inducing acute pancreatitis. CM remnants can be taken up by macrophage surface receptors and are associated with atherosclerosis.
Very Low-Density Lipoproteins (VLDL)
These lipoproteins are synthesized in the liver, with TG accounting for approximately 55% of their content. They, along with CM, are collectively referred to as triglyceride-rich lipoproteins. VLDL primarily transports endogenous TG to extrahepatic tissues while indirectly or directly delivering CH to peripheral tissues. In serum lacking CM, TG concentration reflects VLDL levels. Elevated VLDL levels are a risk factor for coronary heart disease.
Low-Density Lipoproteins (LDL)
These are formed through the hydrolysis of TG from VLDL and IDL. Approximately 50% of LDL content is CH, making it the lipoprotein with the highest cholesterol content and thus referred to as CH-rich lipoprotein. Apo B100 constitutes more than 95% of its apolipoprotein content. LDL primarily transports CH to extrahepatic tissues, where it binds with LDL receptors, mediating CH uptake and utilization. Elevated LDL-C levels are closely correlated with increased cholesterol levels in cases of isolated hypercholesterolemia. LDL is a major risk factor for atherosclerosis. Small, dense LDL (sLDL) particles, classified in subtypes LDL 3–7, readily penetrate arterial walls. Both sLDL and oxidatively modified LDL strongly promote atherosclerosis.
High-Density Lipoproteins (HDL)
These lipoproteins are primarily synthesized in the liver and small intestine, with their protein and lipid content constituting approximately 50% each. The main apolipoproteins are apo A1 and apo A2. HDL is responsible for transporting CH from peripheral tissues to the liver for recirculation or excretion as bile acids, a process known as reverse cholesterol transport. HDL includes multiple subcomponents, with varying anti-atherosclerosis properties. Low HDL-C is an independent risk factor for atherosclerotic cardiovascular disease (ASCVD).
Lipoprotein(a) [Lp(a)]
This is an independent lipoprotein synthesized by the liver, containing apo B100 and apo(a) as apolipoproteins. Serum Lp(a) levels are primarily determined by genetic factors and are inversely correlated with the size of apo(a). Lp(a) is an independent risk factor for ASCVD and calcific aortic valve stenosis.
Classification
Common methods for classifying dyslipidemia include phenotypic classification, etiological classification, and clinical classification, with clinical classification being the most practical.
Phenotypic Classification
The World Health Organization (WHO) classifies dyslipidemia into five types based on the types of lipoproteins involved and the severity of the condition, with Type II further divided into two subtypes. Among these, Types IIa, IIb, and IV are more commonly observed.
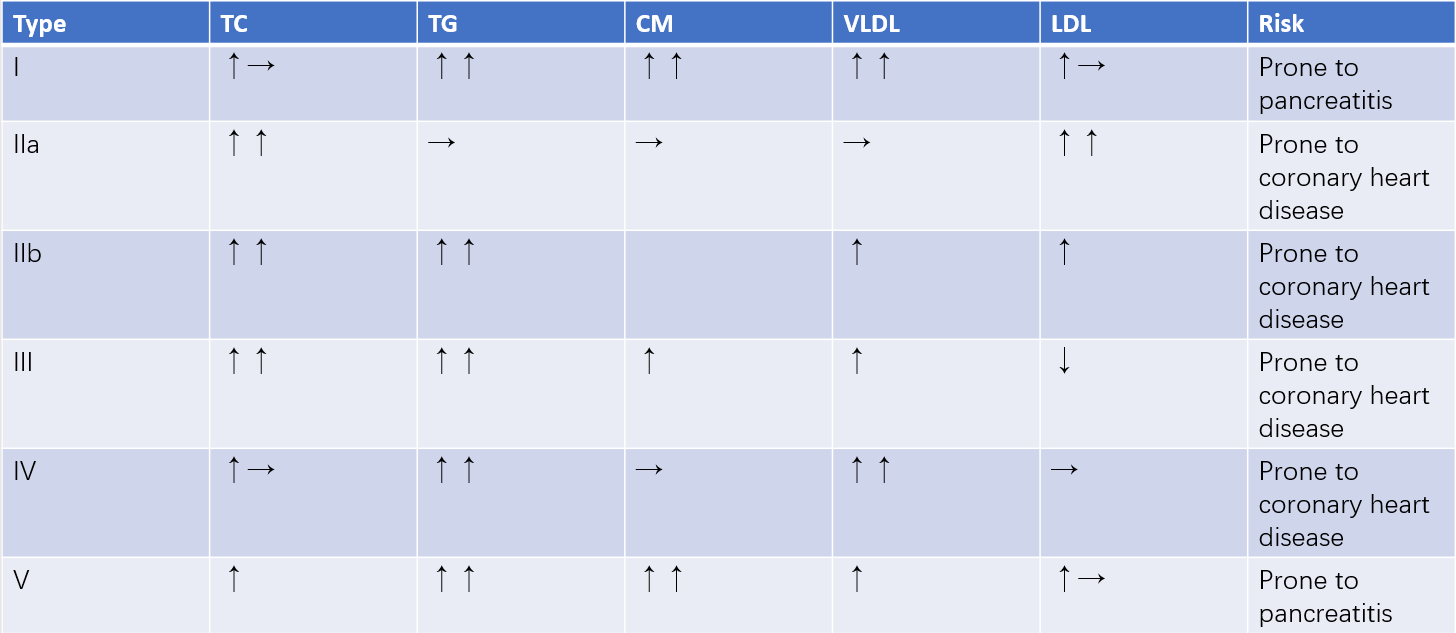
Table 2 Phenotype classification of lipoprotein abnormalities
Note: ↑ indicates elevated levels; → indicates normal levels; ↓ indicates reduced levels.
Etiological Classification
Primary Dyslipidemia
This category typically arises from genetic factors without any identifiable secondary factors such as disease or medication causing the dyslipidemia. It may result from monogenic mutations (e.g., familial hypercholesterolemia, FH) or polygenic mutations (commonly referred to as sporadic or polygenic dyslipoproteinemia).
Secondary Dyslipidemia
This form of dyslipidemia results from underlying systemic diseases, altered metabolic states, unhealthy dietary habits, or certain medications that affect the metabolism of serum lipids and lipoproteins.
Clinical Classification
Clinically, dyslipidemia is classified as hypercholesterolemia, hypertriglyceridemia, mixed hyperlipidemia, or low HDL-C dyslipidemia.
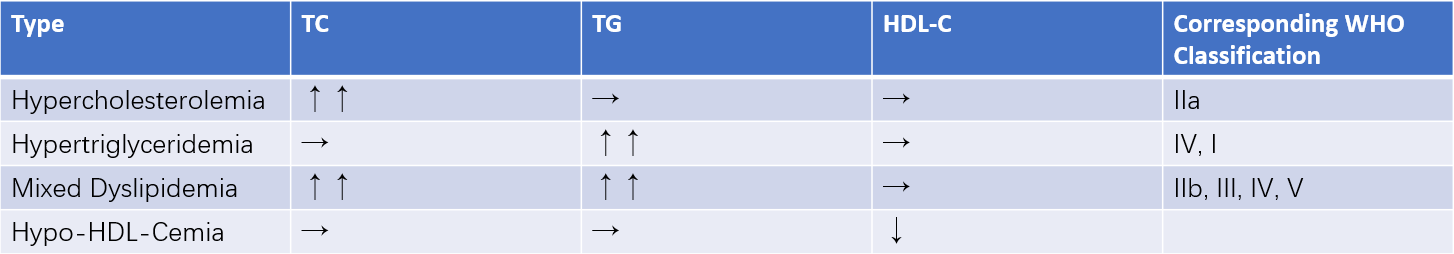
Table 3 Clinical classification of dyslipidemia
Note: ↑ indicates elevated levels; → indicates normal levels; ↓ indicates reduced levels.
Etiology and Pathogenesis
Abnormalities in lipid sources, lipoprotein synthesis, metabolic enzyme activity, or receptor pathway dysfunction during degradation processes can lead to dyslipidemia.
Primary Dyslipidemia
Primary dyslipidemia is mostly caused by mutations in single or multiple genes and often exhibits familial clustering with a clear genetic predisposition.
Familial dyslipoproteinemia results from genetic defects. Familial lipoprotein lipase (LPL) deficiency and familial apo C2 deficiency can impair the degradation of chylomicrons (CM) and very low-density lipoproteins (VLDL), leading to Types I and V dyslipoproteinemia.
Gene mutations associated with familial hypercholesterolemia include loss-of-function mutations in the LDL receptor gene, mutations in the apo B gene encoding for LDL receptor binding, gain-of-function mutations in the PCSK9 gene encoding proprotein convertase subtilisin/kexin type 9 (which breaks down LDL receptors), and mutations in the LDL receptor chaperone gene responsible for translocating LDL receptors to the cell membrane. These primarily manifest as Type II dyslipoproteinemia, with loss-of-function mutations in the LDL receptor gene being the most common cause of familial hypercholesterolemia.
Familial hypertriglyceridemia is often caused by mutations in genes involved in triglyceride metabolism, such as LPL, apo C2, or apo A5.
Secondary Dyslipidemia
Several systemic diseases or conditions, including hypothyroidism, Cushing’s syndrome, diabetes mellitus, liver and kidney diseases, systemic lupus erythematosus, multiple myeloma, and polycystic ovary syndrome, can induce secondary dyslipidemia. These conditions can influence different stages of lipid or lipoprotein synthesis, transport, and metabolism.
Prolonged use of certain medications can also lead to secondary dyslipidemia. For example, thiazide diuretics can elevate serum total cholesterol (TC), TG, VLDL, and LDL levels while reducing HDL levels. Long-term high-dose use of glucocorticoids can promote lipolysis, resulting in elevated plasma TC and TG levels.
Unhealthy diets and alcohol consumption can also contribute. Diets rich in saturated fatty acids and cholesterol can increase cholesterol levels, while excessive intake of simple carbohydrates or alcohol can lead to hypertriglyceridemia.
Clinical Presentation
Dyslipidemia can occur across different ages and genders, with apparent dyslipidemia often being associated with a family history. Blood lipid levels tend to increase with age, peaking around 50–60 years before stabilizing or slightly declining. In younger and middle-aged adults, blood lipid levels in women are lower than those in men, but levels significantly increase after menopause, often exceeding those in men of the same age.
Xanthomas, Premature Corneal Arcus, and Fundus Changes
Xanthomas are abnormal localized elevations of the skin caused by lipid deposits. They can appear yellow, orange, or reddish-brown and often present in the form of nodules, plaques, or papules with a soft texture, typically seen around the eyelids.
Dyslipidemia may result in corneal arcus, a grayish-white or white ring located at the peripheral edge of the cornea, caused by lipid deposition in individuals under the age of 40.
Severe hypertriglyceridemia may lead to lipemia retinalis, causing changes in the fundus.
Atherosclerosis
Lipid deposition in the subendothelial layer of blood vessels can lead to atherosclerosis, which is associated with cardiovascular, cerebrovascular, and peripheral vascular diseases. Familial dyslipidemia may lead to coronary heart disease (CHD) or even myocardial infarction at a young age, often before puberty. Severe hypercholesterolemia can result in migratory polyarthritis. Severe hypertriglyceridemia (>5.6 mmol/L) may lead to acute pancreatitis.
Laboratory Tests
Dyslipidemia is diagnosed and classified through laboratory testing. The primary tests include measurement of plasma or serum total cholesterol (TC), triglycerides (TG), low-density lipoprotein cholesterol (LDL-C), and high-density lipoprotein cholesterol (HDL-C). Blood samples are collected after fasting for 12–14 hours, and the meal prior to blood collection should exclude high-fat foods and alcohol.
Diagnosis and Differential Diagnosis
Diagnosis
A comprehensive medical history, including dietary and lifestyle habits, history of diseases related to secondary dyslipidemia, medication history, and family history, is an essential aspect of the diagnostic process. Physical examinations focus on identifying the presence of xanthomas, corneal arcus, or fundus changes associated with lipemia.
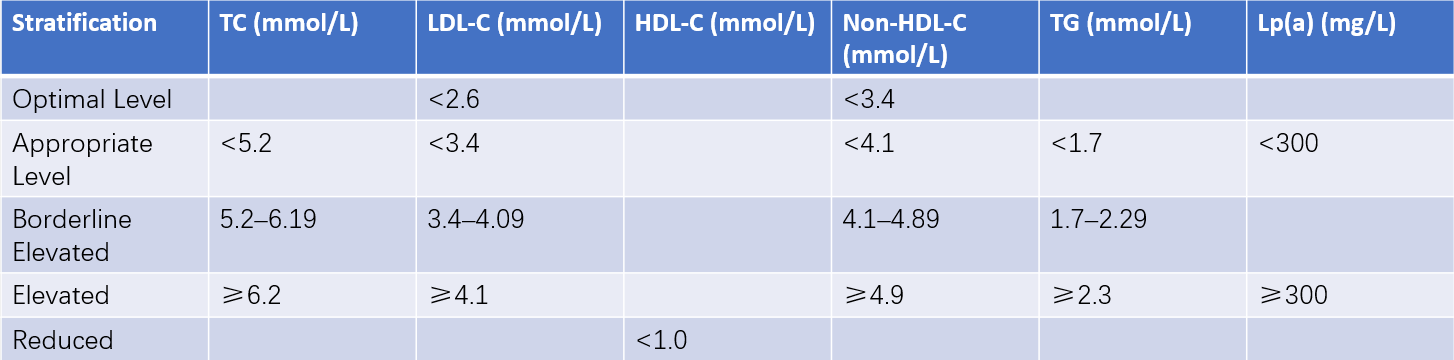
Table 4 Diagnostic and stratification criteria for dyslipidemia
Note: These reference standards apply only to low-risk populations undergoing primary prevention for ASCVD. Listed values correspond to fasting lipid levels measured 12 hours prior to intervention.
Screening
Early detection of dyslipidemia and dynamic monitoring of blood lipid levels are necessary measures for preventing and managing atherosclerotic cardiovascular disease (ASCVD). Adults aged 20–40 are advised to undergo lipid screening at least once every five years, while men over 40 and postmenopausal women should have annual testing. Individuals with ASCVD or those at high risk for ASCVD should have their lipid profiles tested every 3–6 months. When lipid abnormalities are detected for the first time, retesting within 2–4 weeks is required. Persistent abnormalities in subsequent tests confirm the diagnosis.
Key populations for lipid screening include:
- Individuals with a family history of dyslipidemia, coronary heart disease, or atherosclerosis, especially in cases where first-degree relatives have early-onset coronary heart disease or other atherosclerotic conditions.
- Patients with a history of ASCVD.
- Individuals with multiple ASCVD risk factors, including hypertension, diabetes, obesity, excessive alcohol consumption, or a history of smoking.
- Individuals presenting with skin or tendon xanthomas.
Differential Diagnosis
Gene testing may be considered for diagnosing primary dyslipidemia, such as familial dyslipoproteinemia. Secondary dyslipidemia commonly presents with clinical manifestations and pathological features of the underlying primary condition. Particular attention is required for distinguishing secondary dyslipidemia caused by the following disorders:
Hypothyroidism
Hypothyroidism is often associated with dyslipidemia, commonly presenting as Type IIa (isolated hypercholesterolemia) or Type IIb (mixed hyperlipidemia). Reduced thyroid hormone secretion in hypothyroidism leads to decreased LDL-C clearance, increased cholesterol synthesis, and reduced transformation. Thyroid-stimulating hormone (TSH) directly regulates lipid metabolism, promoting cholesterol and TG synthesis while inhibiting cholesterol transformation.
Cushing's Syndrome
Dyslipidemia in the context of Cushing's syndrome most commonly presents as Type IIb (mixed hyperlipidemia).
Nephrotic Syndrome
Hyperlipidemia is a clinical feature of nephrotic syndrome, with elevated levels of TC, LDL-C, small dense LDL (sLDL), apo B, apo C2, apo E, and Lp(a). The main mechanism involves hypoalbuminemia, leading to increased synthesis and reduced degradation of lipoproteins.
Treatment
Treatment Principles
Treatment of Secondary Dyslipidemia
Management of the underlying primary disease is the main focus in secondary dyslipidemia, as conditions like hypothyroidism and diabetes often improve the lipid profile after proper control. Lipid-lowering therapy may be considered if dyslipidemia persists despite the resolution of the primary condition.
Intervention Based on ASCVD Risk Level
Risk assessment is performed based on the presence or absence of ASCVD, dividing patients into primary prevention and secondary prevention categories. Among ASCVD patients, those with two or more major ASCVD events or one major ASCVD event accompanied by two or more high-risk factors are classified as very high-risk individuals, while other ASCVD patients are classified as high-risk. In patients without ASCVD, the following criteria define high-risk individuals:
- LDL-C ≥4.9 mmol/L or TC ≥7.2 mmol/L.
- Diabetic patients aged 40 years or older.
- Chronic kidney disease (CKD) stages 3–4.
For individuals not meeting these conditions, overall 10-year ASCVD risk should be evaluated before deciding on lipid-lowering therapy. Risk is stratified based on 10-year ASCVD incidence rates, categorizing individuals into low risk (<5%), medium risk (5–9%), and high risk (≥10%).
Further lifelong ASCVD risk assessment is recommended for individuals with an intermediate 10-year ASCVD risk and age <55 years to identify high-risk individuals for early intervention. High lifelong ASCVD risk is defined by the presence of two or more of the following factors:
- Systolic blood pressure ≥160 mmHg or diastolic blood pressure ≥100 mmHg.
- Non-HDL-C ≥5.2 mmol/L.
- HDL-C <1.0 mmol/L.
- Body mass index (BMI) ≥28 kg/m2.
- Smoking status.
LDL-C as a Primary Target for Intervention
Elevated LDL-C is the key factor in ASCVD pathophysiology, and lowering LDL-C is the primary goal of intervention strategies. In cases of hypertriglyceridemia where CM remnants are elevated, increased atherosclerosis risk is observed, and non-HDL-C may serve as a secondary target for intervention.
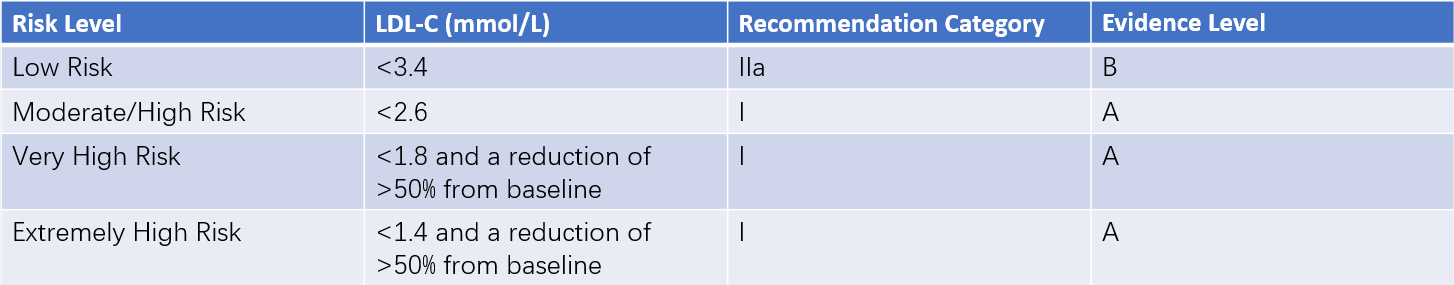
Table 5 Target levels for LDL-C/Non-HDL-C reduction in different ASCVD risk populations
Note: Non-HDL-C target level = LDL-C + 0.8 mmol/L.
Based on overall ASCVD risk stratification, target levels for lipid-lowering interventions should be established. If baseline LDL-C levels are significantly elevated and treatment goals cannot be met, LDL-C should be reduced by at least 50%. For very high-risk and extremely high-risk populations, even if baseline LDL-C levels meet target thresholds, further reduction of LDL-C by 50% is recommended.
Therapeutic Lifestyle Interventions
Therapeutic lifestyle modifications prioritize a healthy lifestyle, including a balanced diet, appropriate increases in physical activity, weight management, and smoking and alcohol cessation. Lifestyle interventions must be consistently maintained regardless of whether medication is chosen.
Balanced Diet
Dietary adjustments are tailored to the patient’s degree and type of dyslipidemia, as well as factors such as gender, age, and physical activity level. Within daily nutritional and total energy requirements, the intake of dietary cholesterol (CH) is advised to remain below 300 mg/day, complemented with plant sterols (2–3 g/day). Saturated fat intake should be limited (generally less than 10% of total energy for the general population and less than 7% for those with hypercholesterolemia), with a preference for foods rich in omega-3 polyunsaturated fatty acids. Carbohydrates should account for 50–60% of total energy, while soluble dietary fiber intake should range from 10–25 g/day.
Increased Physical Activity
Moderate-intensity exercise for 30 minutes per day, 5–7 days per week, is recommended in order to maintain a healthy body mass index (BMI of 20.0–23.9 kg/m²). For ASCVD patients, exercise safety should be thoroughly assessed with exercise stress tests.
Additional Lifestyle Adjustments
Smoking cessation, salt restriction, alcohol moderation, and avoidance of strong alcoholic beverages are also important.
Medication Therapy
Statins
Statins act by competitively inhibiting HMG-CoA reductase, the rate-limiting enzyme in cholesterol synthesis, leading to reduced cholesterol production. They accelerate LDL clearance and inhibit VLDL synthesis. Statins significantly lower serum TC, LDL-C, and apo B while also moderately lowering TG and slightly increasing HDL-C levels.
Statins are indicated for hypercholesterolemia, mixed hyperlipidemia, and ASCVD. Commonly used statins and their daily dosage ranges include:
- Lovastatin (10–80 mg/day)
- Simvastatin (5–40 mg/day)
- Pravastatin (10–40 mg/day)
- Fluvastatin (10–40 mg/day)
- Atorvastatin (10–80 mg/day)
- Rosuvastatin (10–20 mg/day)
Adverse effects are uncommon but may include elevated transaminase levels, myalgia, myositis, and increased serum creatine kinase. Rarely, rhabdomyolysis may occur, potentially leading to acute renal failure. Adjusting the type of statin, reducing the dose, administering on alternate days, or switching to non-statin medications may mitigate these side effects.
Statins should not be used concurrently with ciclosporin, Tripterygium wilfordii, cyclophosphamide, macrolide antibiotics, or azole antifungals (e.g., ketoconazole). Statins are contraindicated in children, pregnant or lactating women, and women planning to conceive. Long-term statin use has been associated with an increased risk of diabetes mellitus.
Proprotein Convertase Subtilisin/Kexin Type 9 (PCSK9) Inhibitors
PCSK9 inhibitors enhance LDL-C clearance by preventing the degradation of LDL receptors. Approved agents include evolocumab and alirocumab. Evolocumab (140 mg) or alirocumab (75 mg) subcutaneous injections are administered once every two weeks. These medications are well-tolerated with good safety profiles, with the most common side effects being injection site itching and flu-like symptoms.
Additionally, small interfering RNA (siRNA) targeting PCSK9 can bind to mRNA encoding PCSK9 protein, utilizing RNA interference to suppress hepatic production of PCSK9 protein and reduce LDL-C levels in the bloodstream. Inclisiran, one such siRNA-based drug, has been approved for clinical use. A single injection can maintain efficacy for six months, making it an ultra-long-acting PCSK9 inhibitor. This drug is generally well-tolerated, with injection site discomfort being the most commonly reported side effect.
Cholesterol Absorption Inhibitors
Ezetimibe inhibits cholesterol absorption by acting on the brush border of intestinal cells. It is suitable for hypercholesterolemia and mixed hyperlipidemia characterized primarily by elevated TC, either as monotherapy or in combination with statins. The recommended dose is 10 mg once daily. Common adverse effects include transient headaches and gastrointestinal symptoms. It is contraindicated during pregnancy and lactation.
Probucol
Probucol promotes LDL clearance via non-receptor pathways, reducing TC and LDL-C. It is particularly indicated for hypercholesterolemia, especially in cases of familial hypercholesterolemia (FH) and xanthomas. The usual dose is 0.5 g orally, twice daily. Common side effects include nausea, with rare occurrences of QT interval prolongation. It is contraindicated in patients with ventricular arrhythmias, QT prolongation, hypokalemia, pregnancy, and lactation.
Bile Acid Sequestrants
Bile acid sequestrants irreversibly bind bile acids in the intestine, interrupt their enterohepatic circulation, and promote their excretion in stool, thereby reducing cholesterol reabsorption. These are suitable for hypercholesterolemia and mixed hyperlipidemia characterized primarily by elevated TC. Common agents and their daily dose ranges include cholestyramine (4–16 g), colestipol (5–20 g), and colesevelam (1.875–4.375 g). Combination therapy with statins can significantly enhance lipid-lowering efficacy. Common side effects include nausea, vomiting, abdominal distension, abdominal pain, and constipation. They may interfere with the absorption of other drugs, such as folic acid, digoxin, fibrates, statins, antibiotics, thyroid hormones, and fat-soluble vitamins. Abnormal β-lipoproteinemia and serum TG > 4.5 mmol/L are absolute contraindications.
Fibrates
Fibrates activate peroxisome proliferator-activated receptor α (PPARα), reducing serum TG and increasing HDL-C levels, while promoting the breakdown of VLDL and TG as well as the reverse transport of cholesterol. They are appropriate for hypertriglyceridemia and mixed hyperlipidemia characterized primarily by elevated TG.
Common agents include fenofibrate (0.1 g, three times daily, or 0.2 g microcrystalline formulation once daily) and bezafibrate (0.2 g, three times daily, or 0.4 g extended-release formulation once daily). Common side effects resemble those of statins. Fibrates enhance the effect of anticoagulants, requiring dose adjustments when used concurrently. They are contraindicated in patients with liver or kidney dysfunction, children, and pregnant or lactating women.
Nicotinic Acid (Niacin) and Derivatives
Nicotinic acid reduces lipase enzyme activity in adipose tissue, lessening the influx of free fatty acids into the liver and diminishing VLDL secretion. It is suitable for hypertriglyceridemia and mixed hyperlipidemia characterized primarily by elevated TG. The recommended dose for nicotinic acid is 1–2 g once daily at bedtime, starting with a small dose (0.375–0.5 g/day) and increasing to the target dose after 4 weeks. Acipimox, a niacin derivative, is dosed at 0.25 g, taken 1–3 times daily after meals. At high doses, nicotinic acid lowers TC, LDL-C, and TG while increasing HDL-C. Common adverse effects include facial flushing, itching, and gastrointestinal symptoms, with occasional liver function impairment or hyperuricemia. Acipimox has fewer side effects.
High-Purity Fish Oil Preparations
The main components of fish oil are omega-3 fatty acids, including eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA). These reduce TG synthesis and secretion while promoting TG clearance from VLDL particles, thereby lowering TG levels. Fish oil is appropriate for hypertriglyceridemia and mixed hyperlipidemia characterized primarily by elevated TG. Adverse effects are rare. It is contraindicated in individuals with a bleeding tendency.
Other Lipid-Lowering Agents
Adenosine Triphosphate Citrate Lyase Inhibitors
Bempedoic acid inhibits cholesterol synthesis and is used for treating heterozygous familial hypercholesterolemia (HeFH) or ASCVD in patients with uncontrolled LDL-C. It demonstrates good overall safety and tolerability.
Angiopoietin-like Protein 3 Inhibitors
Evinacumab binds to angiopoietin-like protein 3, inhibits its function, and enhances fatty acid breakdown and cholesterol clearance. It is approved in the United States for the treatment of homozygous familial hypercholesterolemia (HoFH).
Selection and Combination of Lipid-Lowering Therapies
The choice and combination of lipid-lowering medications should be based on the patient’s type of dyslipidemia, the mechanism of action of the drugs, and their additional pharmacological properties. Combination therapy can improve target lipid achievement rates and reduce the incidence of adverse reactions.
Hypercholesterolemia
Statins are the first choice. If LDL-C targets are not met or statins are not tolerated at moderate intensity, ezetimibe or PCSK9 inhibitors may be considered as combination options. If LDL-C targets remain unmet with dual therapy, triple therapy with statins, ezetimibe, and PCSK9 inhibitors can be employed. This approach enables rapid LDL-C target achievement with good overall safety and cardiovascular benefits.
Hypertriglyceridemia
Fibrates, niacin, or omega-3 fatty acids may be used. Therapeutic lifestyle modifications take precedence when serum TG levels are ≥1.7 mmol/L. For severe hypertriglyceridemia (fasting TG ≥5.6 mmol/L), combinations of fibrates, high-purity omega-3 fatty acids, and niacin may be employed.
Mixed Hyperlipidemia
For cases primarily characterized by elevated TC and LDL-C, statins are preferred. For cases predominantly characterized by elevated TG, fibrates are the first choice. When TC, LDL-C, and TG are all significantly elevated, combination therapy with statins and fibrates or omega-3 fatty acids may be considered. Dual therapy with statins and fibrates can significantly improve the lipid profile but increases the likelihood of adverse effects, making it important to start with a low dose and closely monitor muscle enzymes and liver enzymes. High-dose omega-3 fatty acids, due to their potential for increased bleeding risk, are not recommended for long-term use.
Other Treatment Measures
Lipoprotein Apheresis
Lipoprotein apheresis is an important adjunct treatment for familial hypercholesterolemia (FH) and can reduce LDL-C levels by 55%–70%. The optimal frequency for this treatment is once per week. It is also used for very rare cases of severe refractory hypercholesterolemia in patients who are allergic to or intolerant of statins. This treatment is costly, invasive, and carries a risk of infection.
Surgical Treatment
For extremely severe hypercholesterolemia, such as homozygous familial hypercholesterolemia (HoFH) or severe hypercholesterolemia that is intolerant to medications, surgical approaches may be considered. These include partial ileal bypass surgery, portocaval shunt procedures, and liver transplantation.
Monitoring During the Treatment Process
Lipid-lowering therapy is generally long-term, often lifelong. Individual responses to the same treatment or medication vary widely in terms of efficacy and side effects, making close monitoring of lipid levels and other relevant indicators essential. For patients undergoing non-pharmacological therapy, lipid levels should be re-evaluated after 3–6 months. If targets are achieved, non-pharmacological treatment may be continued, but follow-up assessments every 6–12 months remain necessary.
For patients initiating lipid-lowering medications, lipid profiles, transaminase levels, and creatine kinase should be reassessed within 4–6 weeks. If lipid targets are met and no adverse effects are present, the monitoring intervals may be gradually reduced to every 3–6 months. If lipid targets remain unmet after 1–3 months, medication dosages or types should be adjusted, or combination therapy with lipid-lowering agents having different mechanisms of action may be adopted. Each time the medication type or dosage is adjusted, lipid profiles, transaminase levels, and creatine kinase should be reassessed within 4–6 weeks.
Prevention and Prognosis
Preventive measures for dyslipidemia primarily include promoting health education, encouraging a balanced diet, increasing physical activity and exercise, preventing obesity, and avoiding unhealthy lifestyle habits. These efforts should align with public education campaigns targeting chronic diseases such as obesity, diabetes, and cardiovascular diseases. With active and comprehensive treatment, the prognosis for dyslipidemia is generally favorable.