Rheumatic diseases refer to a group of chronic disorders that affect bones, joints, and surrounding soft tissues such as muscles, tendons, synovium, bursae, ligaments, and cartilage, as well as other related tissues and organs. These diseases comprise over 100 conditions across ten major categories. The etiology of rheumatic diseases is diverse, and their pathogenesis remains poorly understood, though most are closely associated with immune system abnormalities. Rheumatic diseases may involve localized pathological damage or systemic conditions, and without timely diagnosis and treatment, many of these diseases carry the risk of disability or even death, imposing a significant burden on families and society.
The spectrum of rheumatic diseases has changed markedly with advancements in societal development, healthcare standards, and lifestyle habits. Infection-related rheumatic diseases have significantly decreased, while the incidence of osteoarthritis and gouty arthritis has been increasing. With ongoing progress in molecular biology, immunology, and genetics, newer rheumatic conditions such as IgG4-related diseases and autoinflammatory disorders are increasingly recognized. Concurrently, the emergence of novel drugs, including biologics and small-molecule targeting therapies, continues to expand the horizons of rheumatology development.
Classification
The causes and mechanisms of rheumatic diseases are highly complex, and the specific etiology of the majority of these conditions remains unclear. As of now, no comprehensive classification system exists. A widely used classification method in clinical practice is the one established in 1983 by the American Rheumatism Association (ARA), which categorizes rheumatic diseases into ten major groups based on their mechanisms, pathology, and clinical features.
As research progresses, the classification and diagnostic criteria for rheumatic diseases continue to undergo updates and refinements. Over the past decade, many rheumatic diseases, including systemic lupus erythematosus (SLE), rheumatoid arthritis (RA), Sjögren’s syndrome, systemic sclerosis, antiphospholipid syndrome, spondyloarthritis, systemic vasculitis, and inflammatory myopathies, have had their diagnostic (classification) criteria revised—some even more than once. Diagnostic methods have evolved from counting the number of criteria met to calculating weighted scores based on different criteria. These updated standards have greatly advanced early diagnosis and treatment of rheumatic diseases and facilitated more standardized and regulated clinical research.
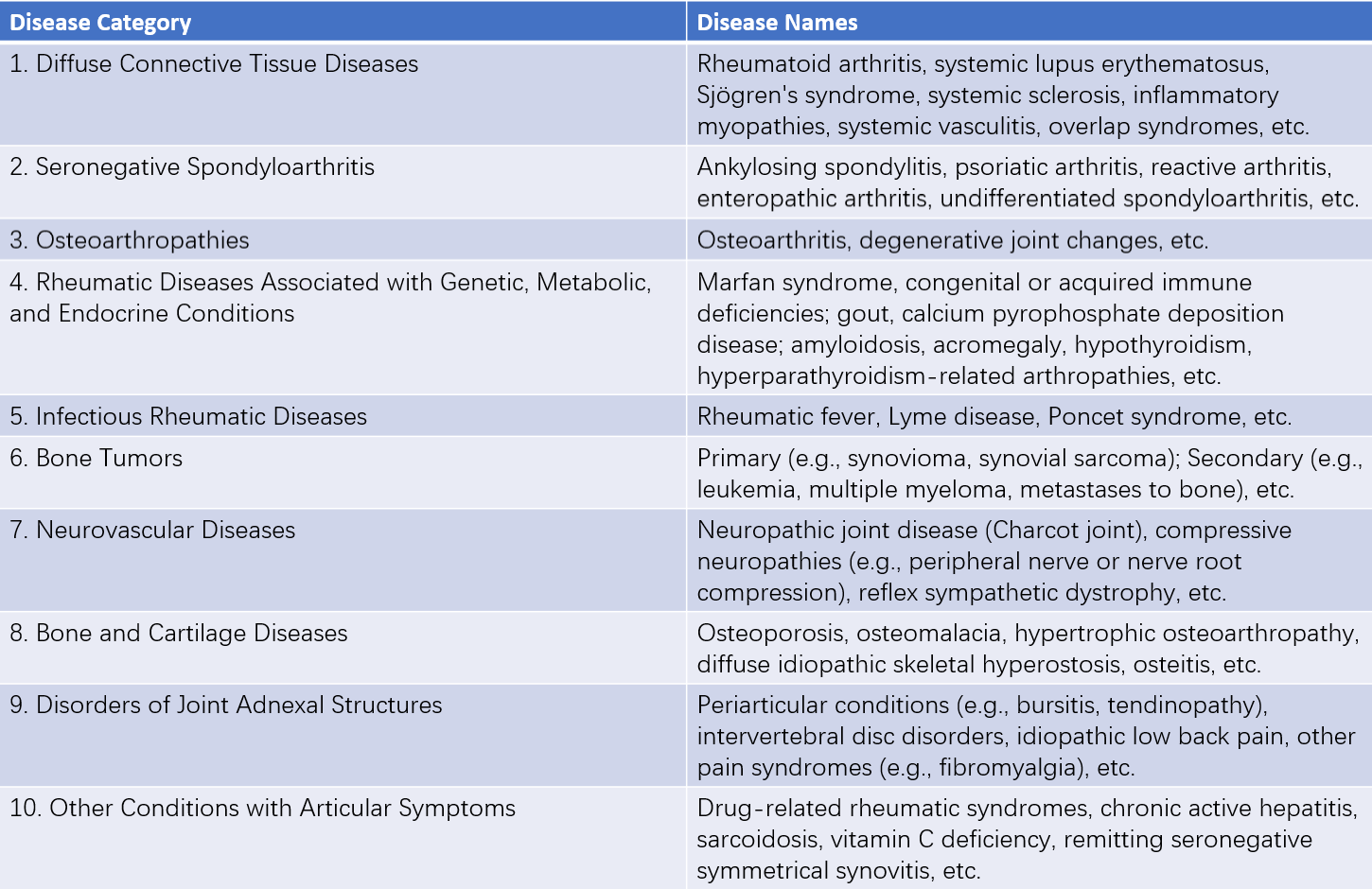
Table 1 Classification and categories of rheumatic diseases
History Taking and Physical Examination
Rheumatic diseases involve multiple disciplines, systems, and organs. Though serologic autoantibody tests and various imaging modalities have greatly enhanced diagnostic precision, meticulous and detailed history taking and physical examination remain central to confirming diagnoses and performing differential diagnoses.
Age of onset, gender, and family history are valuable reference points for diagnosis. For instance, SLE is predominantly observed in women of childbearing age, ankylosing spondylitis (AS) is more common among young men and often has a familial pattern, and osteoarthritis (OA) mainly affects middle-aged and elderly individuals. When collecting history, it is important to inquire beyond the most common symptoms such as bone, joint, and muscle pain, also asking about non-musculoskeletal symptoms such as alopecia, photosensitivity, Raynaud's phenomenon, oral or genital ulcers, dryness of the mouth and eyes, salivary gland swelling, and symptoms from the digestive, respiratory, urinary, nervous, and hematologic systems.
The progression of symptoms often reflects the underlying pathological processes. For patients reporting joint pain, it is crucial to explore the onset pattern, affected locations, number of joints involved, the nature and intensity of the pain, functional status, and its evolution over time. For example, RA typically presents as chronic, symmetric swelling and pain in multiple peripheral joints, with deformity of the joints occurring in later stages.
Physical examination should include not only a general systemic assessment but also inspection of the skin, muscles, and spine and joints. The morphology and distribution of skin lesions may offer diagnostic clues—for instance, malar rash suggests SLE, while purplish-red periorbital swelling and scaly papules on the extensor surfaces of the joints suggest dermatomyositis (DM). Key aspects of muscle examination include assessing for muscle atrophy, tenderness, and weakness. For joint examination, key elements include checking for redness, swelling, tenderness, and range-of-motion limitations in the affected joints.
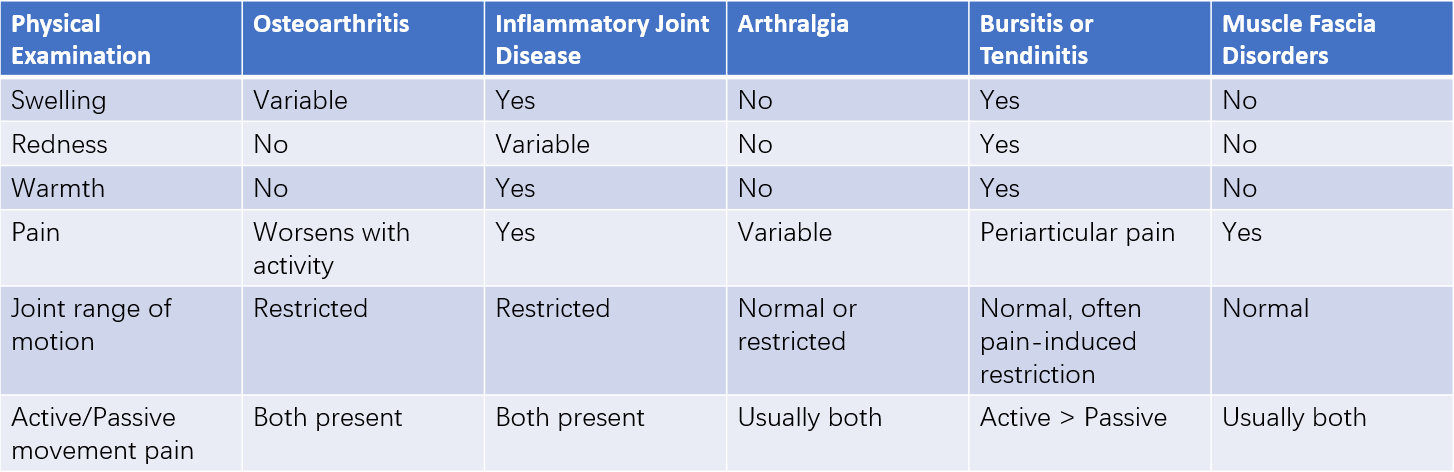
Table 2 Clinical characteristics of joint and periarticular organ lesions
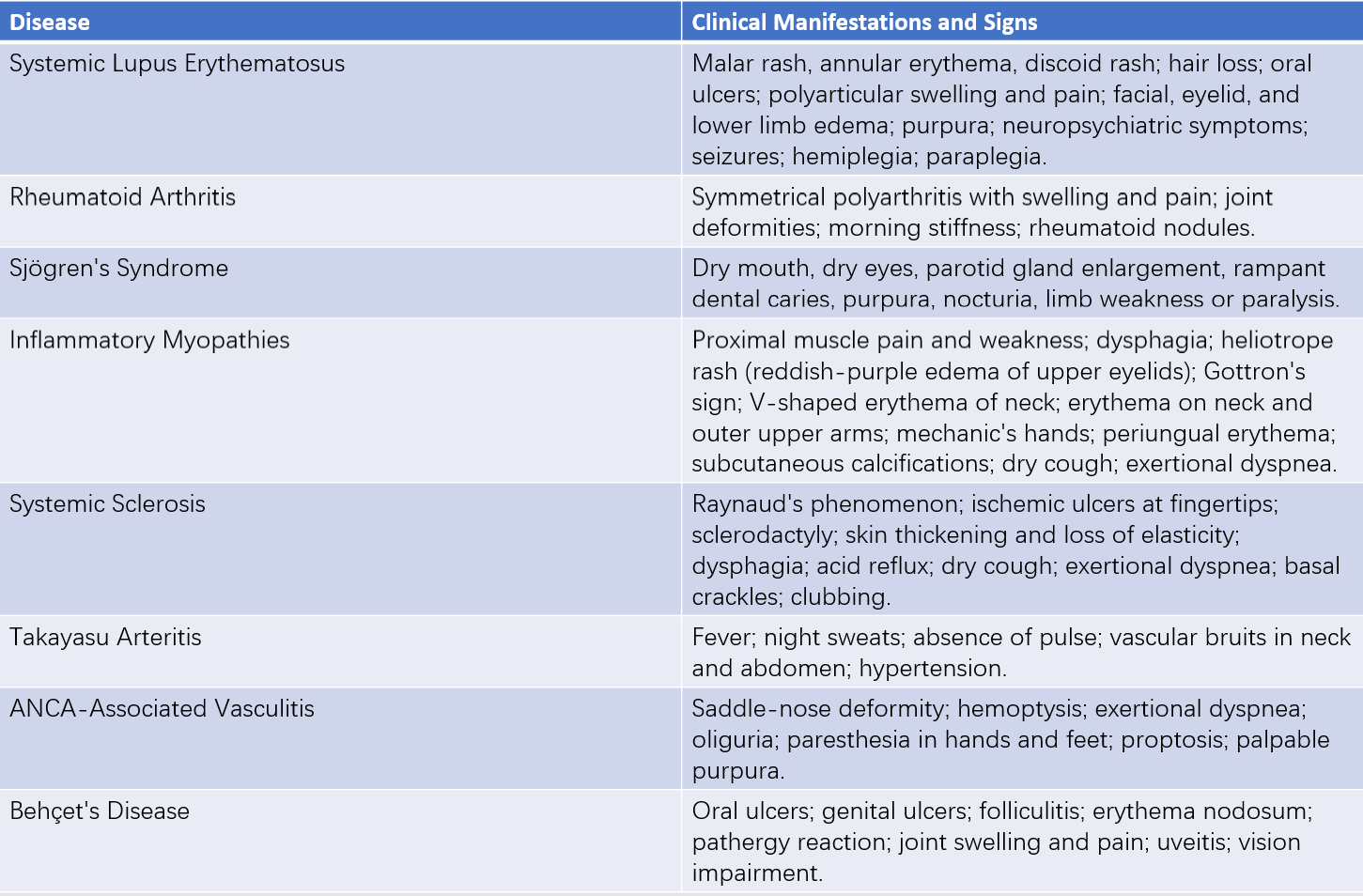
Table 3 Clinical symptoms and signs of common diffuse connective tissue diseases
Laboratory Tests
General Tests
Routine blood, urine, and stool tests, as well as liver and kidney function assessments, are essential. Changes in white blood cell count, hemolytic anemia, thrombocytopenia, proteinuria, and microscopic hematuria may indicate the presence of rheumatic diseases. Tests such as erythrocyte sedimentation rate (ESR), C-reactive protein (CRP), globulin quantification, and complement levels are helpful in diagnosis and in assessing disease activity. For example, elevated ESR and CRP levels are often associated with active rheumatoid arthritis (RA) and vasculitis, while decreased complement C3 and C4 levels often occur during active systemic lupus erythematosus (SLE).
Specific Tests
Autoantibodies
A key characteristic of rheumatic diseases is the presence of autoantibodies in the serum of affected individuals. These are antibodies directed against the body's own tissues, organs, cells, or cellular components. Autoantibody testing plays a significant role in the diagnosis and differential diagnosis of rheumatic diseases. However, the sensitivity and specificity of any autoantibody test are limited, with a certain likelihood of false positives or false negatives. Diagnosis should therefore be based on clinical manifestations rather than antibody test results alone. The main autoantibodies used in clinical rheumatology practice are as follows:
Anti-Nuclear Antibodies (ANA)
ANA targets antigens such as nucleic acids, histones, non-histone proteins, and various proteases. These antigens are found not only in the cell nucleus but also in the cytoplasm and cellular organelles. Accordingly, the scope of ANA targets has expanded from the conventional nucleus to the entire cell. ANA can be categorized into five groups based on the physicochemical properties and distribution of the target antigens: anti-DNA, anti-histone, anti-non-histone, anti-nucleolar, and antibodies against other cellular components. Among these, anti-extractable nuclear antigen (anti-ENA) antibodies, which target a group of soluble antigens extractable with saline, are particularly important for the differential diagnosis of rheumatic diseases, though they do not correlate with disease severity or activity. A positive ANA result may suggest connective tissue disease (CTD), though low-titer ANA can also be found in healthy elderly individuals or in diseases such as cancer. The different ANA components have distinct diagnostic implications and specificities.
Rheumatoid Factor (RF)
RF targets the Fc fragment of denatured IgG molecules, which can form under pathological conditions such as inflammation or when IgG participates in immune responses and undergoes conformational changes. RF positivity is not exclusive to RA and can also occur in primary Sjögren’s syndrome (pSS), SLE, systemic sclerosis (SSc), as well as in infections, malignancies, and approximately 5% of healthy individuals. RF has a positive detection rate of about 80% in RA but is less specific.
Anti-Neutrophil Cytoplasmic Antibodies (ANCA)
ANCA targets various components of neutrophil cytoplasm, notably proteinase 3 (PR3) and myeloperoxidase (MPO), which are closely associated with vasculitis. The presence of ANCA is useful for diagnosing vasculitis.
Anti-Phospholipid Antibodies (APL)
APL targets negatively charged phospholipids, and commonly tested antibodies include anti-cardiolipin antibodies, anti-β2 glycoprotein I antibodies, and lupus anticoagulant. These antibodies are frequently observed in antiphospholipid syndrome (APS) and SLE and primarily cause coagulation system abnormalities. Clinically, they manifest as thrombosis, thrombocytopenia, and pathological pregnancies.
Anti-Keratin Antibody Spectrum
These antibodies target cytoskeletal keratin filaments and are highly specific for RA, aiding in its early diagnosis. Commonly tested markers include anti-perinuclear factor (APF), anti-keratin antibodies (AKA), and anti-cyclic citrullinated peptide (CCP) antibodies. CCP, an artificially synthesized cyclic peptide based on the cDNA sequence of keratin filaments, exhibits better sensitivity and specificity for RA diagnosis compared to AKA.
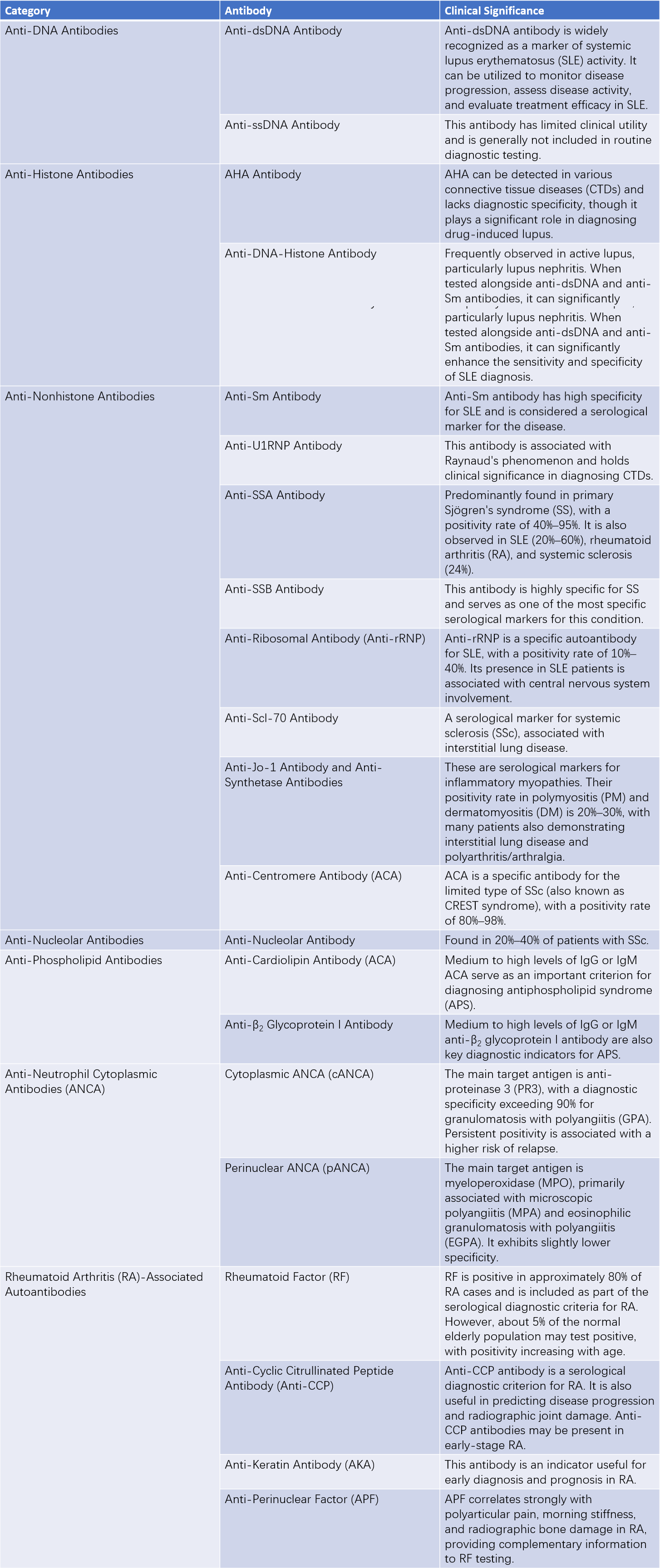
Table 4 Common autoantibodies in antinuclear antibody (ANA) profiles and their clinical significance
Note:
Anti-dsDNA Antibody: Anti-double-stranded DNA Antibody
Anti-ssDNA Antibody: Anti-single-stranded DNA Antibody
SLE: Systemic Lupus Erythematosus
CTDs: Connective Tissue Diseases
SS: Sjögren's Syndrome
SSc: Systemic Sclerosis
PM/DM: Polymyositis/Dermatomyositis
Human Leukocyte Antigen (HLA) Testing
HLA-B27 is closely associated with spondyloarthritis involving axial joints. It has a 90% positive detection rate in ankylosing spondylitis (AS) but is also observed in conditions such as reactive arthritis and psoriatic arthritis, as well as in about 10% of healthy individuals. Additionally, relationships have been noted between HLA-B5 and Behçet's disease (BD), HLA-DR2 and DR3 with SLE, HLA-DR3 and B8 with pSS, and HLA-DR4 with RA.
Synovial Fluid Analysis
Joint fluid can be obtained through joint aspiration, and the white blood cell count in the fluid helps distinguish between inflammatory, non-inflammatory, and septic arthritis. Non-inflammatory arthritis typically presents with white blood cell counts below 2,000×10⁶/L. A count exceeding 3,000×10⁶/L, with more than 50% neutrophils, suggests inflammatory arthritis. Septic arthritis is indicated by purulent joint fluid and even higher white blood cell counts. The presence of urate crystals or positive bacterial smear/culture in the synovial fluid aids the diagnosis of gouty arthritis and infectious arthritis, respectively.
Histopathology
Histopathological findings from biopsy samples provide definitive diagnostic evidence and guide treatment strategies. Renal biopsy aids in the pathological classification of lupus nephritis; synovial biopsy helps determine the causes of arthritis; labial gland biopsy supports the diagnosis of Sjögren’s syndrome; and muscle biopsy is important for diagnosing polymyositis and dermatomyositis.
Imaging Studies
Imaging serves as a critical auxiliary diagnostic tool, contributing to the diagnosis, differential diagnosis, disease staging, and assessment of treatment efficacy for various joint and spinal disorders. Additionally, it aids in evaluating the involvement of organs outside the musculoskeletal system. X-ray imaging is the most commonly used technique for assessing bones and joints. It facilitates diagnosis, differential diagnosis, and long-term follow-up by identifying changes such as soft tissue swelling and calcification, bone demineralization, joint space narrowing, joint erosion or dislocation, and subchondral cystic changes.
Joint CT imaging is particularly valuable for detecting lesions in areas with overlapping tissues, such as the sacroiliac joint, femoral head, sternoclavicular joint, and intervertebral discs. It offers higher sensitivity than X-ray imaging. In recent years, dual-energy CT has been introduced, which enables the identification of gouty tophi in patients with gouty arthritis.
MRI provides distinct imaging features for bones, cartilage, and surrounding tissues, including muscles, ligaments, tendons, and synovium. It is a sensitive and reliable method for detecting soft tissue and articular cartilage injuries, bone marrow edema, ischemic osteonecrosis, early-stage microfractures, and muscle inflammation.
Over the past decade, ultrasound has gained increasing significance in joint evaluation. It allows for the early detection of synovial and cartilage damage and helps monitor disease progression.
Imaging is also crucial for evaluating the involvement of other affected organs. For instance, high-resolution CT of the chest is used to diagnose interstitial lung disease. Cranial CT and MRI play a role in assessing central nervous system involvement in SLE. Techniques such as vascular ultrasound, CT angiography (CTA), magnetic resonance angiography (MRA), conventional angiography, and even positron emission tomography (PET) are helpful for the evaluation of vasculitis.
Treatment
Rheumatic diseases encompass a wide range of conditions, most of which are chronic in nature. Early initiation of treatment following a confirmed diagnosis is essential. The objectives of treatment include maintaining joint and organ function, alleviating symptoms, improving quality of life, and enhancing prognosis. Treatment approaches can be categorized into general management (education, lifestyle changes, physical therapy, exercise, and symptomatic care), pharmacological therapy, and surgical interventions (such as corrective procedures, synovectomy, and joint replacement). The primary drug treatments for rheumatic diseases consist of nonsteroidal anti-inflammatory drugs (NSAIDs), glucocorticoids (GCs), disease-modifying anti-rheumatic drugs (DMARDs), and biological agents. The types and principles of these therapies are outlined as follows:
Nonsteroidal Anti-Inflammatory Drugs (NSAIDs)
NSAIDs exert their effects by inhibiting cyclooxygenase (COX), thereby suppressing the conversion of arachidonic acid into prostaglandins, which leads to anti-inflammatory, antipyretic, and analgesic effects. These drugs are widely used, act quickly, and are effective in pain relief. However, they do not arrest the progression of the underlying disease. Potential adverse effects include impacts on the gastrointestinal tract, kidneys, and cardiovascular system. Clinical use necessitates follow-up monitoring, particularly in elderly patients or those with pre-existing gastrointestinal and renal conditions. Selective COX-2 inhibitors reduce gastrointestinal side effects.
Glucocorticoids (GCs)
Glucocorticoids have strong anti-inflammatory and immunosuppressive properties, making them widely used in the treatment of rheumatic diseases and a first-line therapy for many connective tissue diseases (CTDs). Glucocorticoids can be classified based on their half-life into short-acting (e.g., cortisone, hydrocortisone), intermediate-acting (e.g., prednisone, prednisolone, methylprednisolone, triamcinolone), and long-acting agents (e.g., dexamethasone, betamethasone). Hydrocortisone, prednisolone, and methylprednisolone are hydroxylated at the 11th position and can exert direct physiological effects without liver metabolism, making them preferable for patients with impaired liver function. Long-term and high-dose use of glucocorticoids is associated with numerous adverse effects, including infections, hypertension, hyperglycemia, osteoporosis, rebound effects after withdrawal, aseptic femoral head necrosis, obesity, mood elevation, gastrointestinal ulcers, and others. Clinical application requires careful evaluation of therapeutic benefits versus side effects, with strict consideration of indications, dosage, and duration of treatment, as well as regular monitoring for adverse reactions.
Disease-Modifying Anti-Rheumatic Drugs (DMARDs)
DMARDs are categorized into conventional and targeted DMARDs. These drugs share the ability to modify disease progression and delay structural joint damage, particularly in conditions such as rheumatoid arthritis (RA). DMARDs act slowly, with effects typically observed 2–4 months after initiation. Long-term maintenance therapy is often required once the disease enters remission.
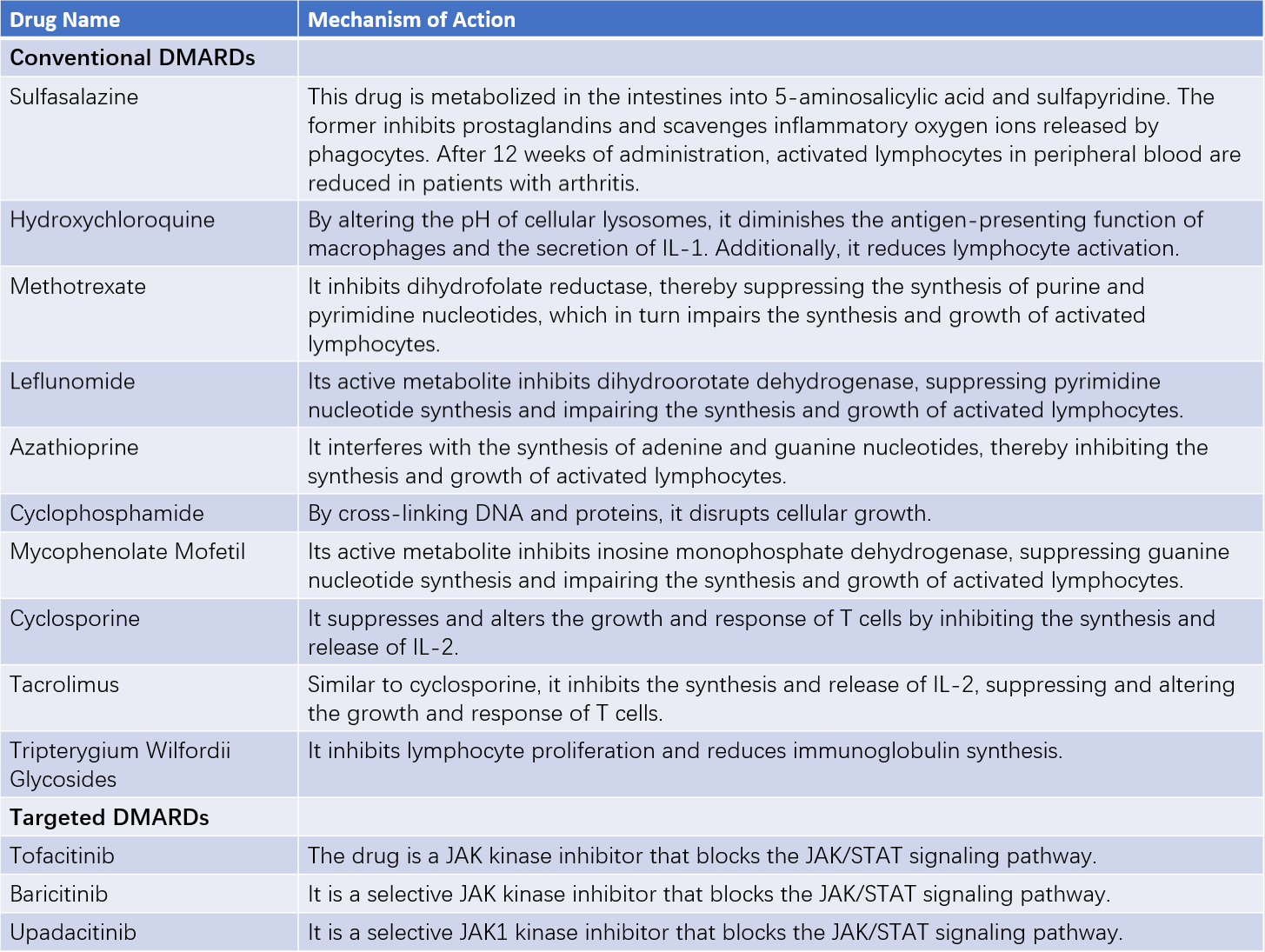
Table 5 Mechanisms of action of DMARDs
Biological Agents
Biological agents, which are monoclonal antibodies produced through genetic engineering, represent one of the most significant advancements in rheumatology over the past three decades. These medications are widely used in the treatment of RA, spondyloarthritis, SLE, systemic vasculitis, and other conditions. Acting through the targeted blockade of specific pathways critical to disease pathogenesis, these agents have revolutionized therapeutic strategies.
Among biological agents, anti-tumor necrosis factor-alpha (TNF-α) drugs were the first to achieve success in treating RA and spondyloarthritis. These agents rapidly improve symptoms, prevent joint destruction, and enhance joint function. Interleukin-6 (IL-6) receptor antagonists and the fusion protein CTLA-4 linked to the Fc fragment of IgG1 (abatacept) are effective for treating RA. Rituximab, an anti-CD20 monoclonal antibody, has been approved for first-line treatment of ANCA-associated vasculitis and has shown promising results in managing refractory SLE, particularly lupus nephritis, hemolytic anemia, and thrombocytopenia. Belimumab, an anti-B cell activating factor monoclonal antibody, and telitacicept are used in SLE patients who do not respond adequately to standard treatments. Complement pathway inhibitors and cell-based therapies also hold potential applications in the management of rheumatic diseases.
The rapid development of biological agents has positioned them as an integral part of the pharmacological arsenal against rheumatic diseases. However, the most common adverse effects include infections and hypersensitivity reactions. Clinical use requires strict adherence to indications and thorough screening for infections, especially hepatitis B and tuberculosis, to prevent severe adverse events.
Adjunctive Therapies
Intravenous immunoglobulin therapy, plasma exchange, and plasma immunoadsorption have demonstrated certain levels of efficacy and can serve as supplementary treatments for some rheumatic autoimmune conditions.