Acute organic phosphorus insecticide poisoning (AOPIP) refers to the inhibition of acetylcholinesterase (AChE) activity by organic phosphorus insecticides (OPIs) entering the body, resulting in the accumulation of acetylcholine (Ach) at physiological effect sites. This accumulation leads to muscarinic, nicotinic, and central nervous system toxic symptoms and signs. Patients often succumb to respiratory failure. OPIs are organic phosphate esters or thiophosphate ester compounds, typically appearing as oily liquids ranging in color from pale yellow to brown. They have a slight volatility and a garlic-like odor. Most OPIs, excluding dichlorvos, are poorly soluble in water and organic solvents, are stable in acidic environments, and decompose and lose efficacy in alkaline conditions. Certain OPIs, such as thimet and parathion-methyl, are resistant to alkaline conditions, while dichlorvos becomes even more toxic when exposed to alkali. OPIs are commonly available in forms such as emulsifiable concentrates, oils, and powders.
OPI Classification and Etiology
The toxicity of OPIs varies significantly depending on their substituents. The classification of OPI toxicity, based on the median lethal dose (LD50) of acute oral exposure in rats, is divided into four categories. This classification serves as an important reference for the effective management of OPI poisoning.
- Extremely Toxic: LD50 <10 mg/kg. Examples include thimet (3911), demeton (1059), parathion (1605), mevinphos, and tetraethyl diphosphate (tetron, TEPP).
- Highly Toxic: LD50 ranging between 10–100 mg/kg. Examples include methylparathion, methamidophos, omethoate, dichlorvos (dichlorphos, DDVP), phosphamidon, monocrotophos, isocarbophos, methidathion, and oxydemeton-methyl.
- Moderately Toxic: LD50 ranging between 100–1000 mg/kg. Examples include dimethoate (rogor), fenthion (bensulide), dichlofenthion, ethion (1240), dipterex (trichlorfon), acephate, diazinon, and phosmet.
- Low Toxicity: LD50 ranging between 1000–5000 mg/kg. Examples include malathion (karbofos, maldison, 4049), phoxim, methylacetophos, iodfenphos, phosphorus chloride, and bromophos.
Etiology
Occupational Poisoning
Occupational poisoning primarily occurs during the production process, particularly while refining, discharging, or packaging the insecticides. Poisoning may result from damaged gloves, contaminated clothing, or masks, as well as exposure due to leaks, spills, or absorption through the hands, skin, or inhalation.
Poisoning During Use
Usage-related poisoning occurs when spray applicators experience skin contamination or when pesticide solutions seep through clothing and are absorbed through the skin. Inhalation of airborne OPIs during application or hand contamination with concentrated solutions during preparation can also lead to poisoning.
Poisoning in Daily Life
Poisoning in daily life may occur due to intentional ingestion, accidental ingestion, or consumption of OPI-contaminated food or water. Misuse of OPIs for treating skin conditions or as insect repellents can also result in poisoning.
Toxin Metabolism
Organic phosphorus insecticides (OPIs) are primarily absorbed through the gastrointestinal tract, respiratory tract, and skin mucosa. Once absorbed, they are rapidly distributed throughout the body's organs, with the highest concentration found in the liver, followed by the kidneys, lungs, and spleen. Muscles and the brain exhibit the lowest levels of OPI concentration. OPIs undergo biotransformation and metabolism primarily in the liver. Some OPIs become more toxic after oxidation. For example, parathion is oxidized into paraoxon via the microsomal oxidase system in liver cells, where paraoxon exhibits an inhibitory effect on cholinesterase (ChE) that is 300 times stronger than parathion. Similarly, demeton undergoes oxidation to form sulfoxide, which increases its ChE inhibition by fivefold. The hydrolysis of OPIs reduces their toxicity. In the liver, trichlorfon undergoes dehydrochlorination to form dichlorvos, which exhibits greater toxicity before being detoxified further via hydrolysis, deamination, and dealkylation processes. Malathion becomes detoxified through ester hydrolysis within the liver. Blood concentrations of OPIs peak 6 to 12 hours after absorption, and urinary excretion through the kidneys typically occurs within 24 hours, with complete elimination achieved within 48 hours.
Upon entering the body, OPIs rapidly bind with ChE, forming stable phosphorylated cholinesterase, which results in a loss of ability to hydrolyze acetylcholine (Ach). This leads to excessive accumulation of Ach at nerve terminals, causing overstimulation of cholinergic neurons and a series of muscarinic-like, nicotinic-like, and central nervous system symptoms.
Mechanism of Toxicity
While OPIs inhibit various enzymes, their main toxic effect in humans and animals revolves around ChE inhibition. ChE in the body is divided into two types: genuine cholinesterase (or acetylcholinesterase, AChE) and pseudocholinesterase (or butyrylcholine esterase). Genuine ChE predominantly exists in the brain's gray matter, red blood cells, sympathetic ganglia, and neuromuscular junctions, where it demonstrates the highest activity in hydrolyzing Ach.
PseudoChE is present in brain white matter's neuroglial cells, plasma, liver, kidneys, submucosa of the intestines, and some glands. It can hydrolyze butyrylcholine but has limited ability to hydrolyze Ach. In cases of severe liver damage, pseudoChE activity is reduced. Genuine ChE inhibited by OPIs recovers relatively quickly at nerve terminals, with most of the inhibited portion restoring function by the second day. However, inhibited red blood cell ChE generally cannot recover on its own until new red blood cells are regenerated, which may take several months. PseudoChE exhibits greater sensitivity to OPIs but recovers more rapidly after inhibition.
The toxic action of OPIs stems from their binding to the esterase site of genuine ChE, forming stable phosphorylated cholinesterase. This inhibits ChE's ability to hydrolyze Ach, triggering excessive accumulation of Ach and resulting in muscarinic, nicotinic-like, and central nervous system symptoms. Severe cases often culminate in respiratory failure. Chronic OPI exposure significantly suppresses ChE activity, although clinical symptoms may remain mild, possibly due to the body's enhanced tolerance to accumulated Ach.
Clinical Manifestations
Acute Poisoning
The onset time and symptoms of acute poisoning are closely related to factors such as the type of toxin, dosage, route of entry, and condition of the body (e.g., fasting versus post-meal). Oral poisoning manifests within 10 minutes to 2 hours, inhalation-induced poisoning within a few minutes to half an hour, and skin absorption-related poisoning within approximately 2 to 6 hours. Acute poisoning can affect individuals, families, or groups and is characterized by an acute cholinergic crisis, which includes the following:
Muscarinic Symptoms (also referred to as M-like symptoms)
These symptoms are primarily caused by overstimulation of parasympathetic nerve terminals, mimicking muscarinic effects. Key manifestations include:
- Smooth muscle spasms resulting in symptoms such as miosis, abdominal pain, and diarrhea.
- Sphincter relaxation leading to involuntary urination and defecation.
- Increased glandular secretion, such as excessive sweating, tearing, and salivation.
- Increased airway secretions, presenting as coughing, dyspnea, and difficulty breathing, often accompanied by dry or moist crackles in both lungs. Severe cases may lead to pulmonary edema.
Nicotinic Symptoms (also referred to as N-like symptoms)
Caused by excessive Ach accumulation at the neuromuscular junction of striated muscles, these symptoms include:
- Muscle tremors and generalized tonic spasms.
- Potential muscle weakness or paralysis, leading to respiratory muscle paralysis and subsequent respiratory failure or cessation.
- Release of catecholamines from postganglionic sympathetic nerve terminals, resulting in elevated blood pressure and cardiac arrhythmias.
Central Nervous System Symptoms
When blood AChE activity is significantly reduced but brain AChE activity remains above 60%, central symptoms and signs are typically absent. However, when brain AChE activity falls below 60%, individuals may exhibit dizziness, headaches, agitation, delirium, seizures, and coma. In severe cases, respiratory and circulatory failure may occur, leading to death.
Local Damage
Certain OPIs can cause allergic dermatitis, skin blistering, or exfoliative dermatitis when in contact with the skin. Eye contamination may result in conjunctival hyperemia and miosis.
Delayed Polyneuropathy
Delayed polyneuropathy may occur 2–3 weeks after symptoms subside in patients with acute severe or moderate OPI poisoning (such as poisoning caused by methamidophos, dichlorvos, dimethoate, or trichlorfon). It manifests as a sensory and motor polyneuropathy, primarily affecting the distal limbs, and may lead to lower limb paralysis and muscle atrophy in the extremities. Current understanding suggests that this condition is not caused by ChE inhibition but may result from OPI inhibition of neuropathy target esterase (NTE), leading to its aging. Whole blood or red blood cell ChE activity is normal in such cases. Electromyography often reveals neurogenic damage.
Intermediate Syndrome
Intermediate syndrome (IMS) typically occurs 24–96 hours after severe OPI poisoning, such as poisoning caused by methamidophos, dichlorvos, or monocrotophos, particularly in cases where inadequate amounts of ChE reactivators have been used. This syndrome emerges after the resolution of acute cholinergic crises following treatment, when the patient is conscious or prior to the onset of delayed polyneuropathy. It is characterized by sudden weakness of the flexor muscles of the neck, proximal limb muscles, and muscles controlled by cranial nerves III, VII, IX, and X. Symptoms include ptosis, ophthalmoplegia, facial paralysis, and respiratory muscle paralysis, leading to ventilatory impairment, respiratory difficulty, or failure, which may be fatal. This condition is associated with prolonged ChE inhibition, which impairs post-synaptic neuromuscular junction function. Whole blood or red blood cell ChE activity is typically below 30%. High-frequency repetitive nerve stimulation often shows progressively diminishing amplitude.
Laboratory Examinations
Measurement of Blood ChE Activity
Blood ChE activity serves as a specific laboratory marker for diagnosing OPI poisoning and is critical for assessing the severity, therapeutic efficacy, and prognosis of poisoning. Blood ChE activity is expressed as a percentage of normal levels, with 100% representing normal activity in a healthy individual. In cases of acute OPI poisoning, ChE activity between 50%–70% indicates mild poisoning, 30%–50% indicates moderate poisoning, and below 30% indicates severe poisoning. For individuals with long-term OPI exposure, blood ChE activity measurement may be used as a biochemical monitoring indicator.
Toxin Detection
OPIs or their specific metabolites can be detected in the blood, urine, feces, or gastric contents of patients. In the body, parathion and methyl parathion are oxidized to p-nitrophenol, while trichlorfon is metabolized into trichloroethanol. The presence of p-nitrophenol or trichloroethanol in the urine supports the diagnosis of poisoning caused by these substances. Dynamic monitoring of OPI blood concentrations is valuable for assessing the severity and progression of acute OPI poisoning (AOPIP) and guiding treatment.
Diagnosis
The diagnosis is based on the following criteria:
- A history of exposure to OPIs.
- Symptoms and signs consistent with OPI poisoning, particularly the presence of a garlic-like odor in exhaled breath, miosis, excessive sweating, pulmonary edema, muscle fasciculations, and coma.
- Decreased ChE activity to varying degrees in whole blood.
- Detection of OPIs or their metabolites in blood or gastric contents.
Additional considerations during diagnosis include the observation that patients with dimethoate or malathion poisoning may experience a sudden worsening of symptoms or even acute OPI poisoning symptoms and sudden death several days to a week after apparent improvement. This "rebound" phenomenon may be associated with delayed reabsorption of residual OPIs in the body or premature discontinuation of antidotes.
Differential Diagnosis
OPI poisoning should be differentiated from conditions such as heatstroke, acute gastroenteritis, or encephalitis. It is also necessary to distinguish it from pyrethroid poisoning (characterized by red papules or blister-like lesions on the skin, with normal blood ChE activity) and formamidine poisoning (characterized by cyanosis, mydriasis, and hemorrhagic cystitis).
Classification of Acute Poisoning Diagnosis
Mild poisoning is characterized solely by muscarinic-like (M-like) symptoms, with ChE activity between 50%–70%.
Moderate poisoning is marked by more severe M-like symptoms accompanied by nicotinic-like (N-like) symptoms; ChE activity is between 30%–50%.
Severe poisoning is defined by the presence of both M-like and N-like symptoms, along with pulmonary edema, convulsions, coma, respiratory muscle paralysis, and cerebral edema; ChE activity is below 30%.
Treatment
Rapid Elimination of the Toxin
The patient needs to be removed from the toxic environment. Toxins that have not yet been absorbed into the bloodstream require thorough removal. Contaminated clothing can be taken off, and skin, hair, and nails can be washed with soap and water. If the eyes are affected, they can be irrigated with clean water, saline, 2% sodium bicarbonate solution, or 3% boric acid solution. For oral poisoning, gastric lavage can be performed with clean water, 2% sodium bicarbonate solution (not suitable for trichlorfon poisoning), or 1:5000 potassium permanganate solution (not suitable for parathion poisoning). This procedure can be repeated multiple times every 3–4 hours, ensuring the lavage returns clear. Subsequently, 20–40 g of sodium sulfate dissolved in 20 ml of water can be administered orally. If no cathartic effect is observed within 30 minutes, additional water (up to 500 ml) can be introduced orally or via a nasogastric tube.
Emergency Resuscitation
Patients with OPI poisoning often succumb to complications such as pulmonary edema, respiratory muscle paralysis, or respiratory failure. It is essential to implement emergency resuscitation measures for such patients. Respiratory secretions can be cleared, and airway patency can be maintained with oxygen therapy or mechanical ventilation based on the patient's condition. Atropine may be used for pulmonary edema as other agents like aminophylline and morphine should be avoided. In cases of cardiac arrest, external chest compressions and other resuscitation techniques are indicated.
Antidotes
Antidotal therapy involves the concurrent use of cholinesterase-reactivating agents and cholinoceptor-blocking drugs.
Principles of Drug Administration
A timely, adequate, combined, and repeated application of antidotes may be required. Appropriate administration routes and optimal timing for discontinuation of the antidotes are considered based on the patient’s condition. Combining cholinoceptor antagonists and cholinesterase reactivators early in poisoning can result in better therapeutic outcomes.
Cholinesterase Reactivators
Oxime compounds can restore the activity of inhibited ChE. The mechanism lies in the positively charged quaternary ammonium nitrogen in the pyridine ring of oxime compounds, which is attracted to the anionic site of phosphorylated cholinesterase. The oxime group interacts with phosphorus in phosphorylated cholinesterase, leading to the release of the enzyme’s ester site and restoring the activity of genuine cholinesterase. These reactivators can also act on peripheral N2 receptors, counteracting peripheral nicotinic cholinoceptor activity and alleviating nicotinic-like toxicity, although they have minimal effects on muscarinic symptoms and central respiratory depression. The commonly used agents include:
Pralidoxime Chloride (PAM-Cl)
This drug exhibits strong reactivation effects, low toxicity, and high water solubility, making it suitable for intravenous or intramuscular injection as the first-line antidote in clinical practice. The initial dosage should be adequate. Indicators of sufficient dosing include the disappearance of peripheral nicotinic-like symptoms (e.g., muscle fasciculations) and restoration of blood ChE activity to 50%–60% or higher. In cases with thorough gastric lavage and mild poisoning, repeat dosing may not be required. Moderate poisoning may need 1–2 additional doses after the initial dose. In severe poisoning, if no indications of sufficient dosage are observed within 30–60 minutes after the first administration, repeated dosing becomes necessary. In cases of large oral doses of dimethoate poisoning, prolonged coma, poor response to cholinesterase reactivators, or significantly decreased blood ChE activity, higher doses of antidotes over 5–7 days may be necessary. Treatment can be stopped when symptoms resolve and blood ChE activity remains above 50%–60%.
Pralidoxime Iodide (PAM-I)
This agent has weaker reactivation effects, lower toxicity, and poor water solubility and is suitable only for intravenous injection. It is considered a second-line antidote in clinical practice.
Obidoxime (DMO4)
Obidoxime demonstrates strong reactivation effects but has higher toxicity. It is highly water-soluble and can be administered intravenously or intramuscularly. ChE reactivators are effective for poisoning caused by substances such as terbufos, demeton, parathion, methamidophos, ethion, and phoxim but are less effective for dichlorvos and trichlorfon poisoning and show limited efficacy for dimethoate and malathion poisoning. However, obidoxime shows better results than pralidoxime iodide for dichlorvos and trichlorfon poisoning.
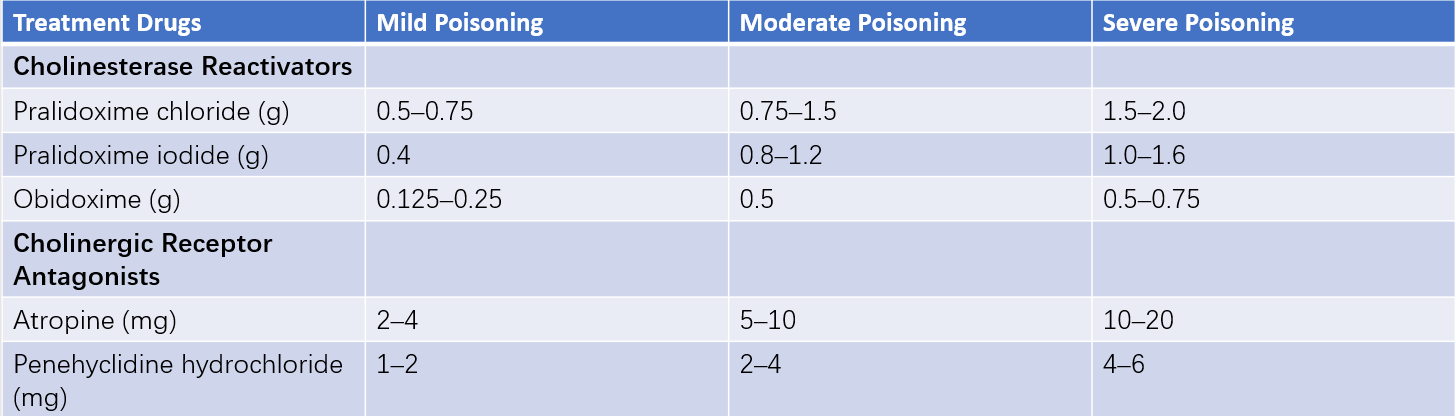
Table 1 Medication for OPI poisoning patients
ChE reactivators are ineffective against ChE that has aged 24–48 hours after poisoning. For patients with poor responses to ChE reactivators, cholinoceptor antagonists may be added.
The adverse effects of ChE reactivators include transient dizziness, blurred vision, diplopia, and increased blood pressure. Large doses can induce seizure-like activity and further inhibit ChE activity. High doses of pralidoxime iodide may cause a bitter taste, dry throat, and nausea. Rapid injection can result in transient respiratory depression. Obidoxime is associated with more noticeable adverse effects, including numbness and burning sensations around the mouth, limbs, or body, nausea, vomiting, facial flushing, and, at excessive doses, ventricular premature contractions, conduction block, or toxic hepatitis.
Cholinoceptor Blocking Drugs
Cholinoceptors are classified into M and N types. M receptors have three subtypes: M1 (found in lung tissue), M2 (found in myocardium), and M3 (found in smooth muscle and glands). N receptors consist of two subtypes: N1 (found in ganglia and postganglionic neurons) and N2 (found in skeletal muscle).
During OPI poisoning, accumulated Ach initially stimulates central N receptors, which then rapidly undergo desensitization, rendering them less responsive to further Ach stimulation. Desensitized N receptors can alter the conformation of M receptors, making them more sensitive to Ach and reducing the efficacy of M receptor antagonists such as atropine. As a result, combining peripheral and central anticholinergic drugs can have synergistic effects.
Muscarinic Cholinergic Receptor Antagonists
These are also known as peripheral anticholinergic agents, including atropine, anisodamine, and penehyclidine hydrochloride. The administration of atropine is performed every 10–30 minutes or 1–2 hours depending on the severity of the condition (refer to Table 9-2-5) until muscarinic symptoms resolve or "atropinization" occurs. Indicators of atropinization include dry mouth, dry skin, increased heart rate (90–100 beats per minute), and resolution of pulmonary rales. At this stage, the dosage of atropine is reduced or discontinued. Symptoms such as significant pupil dilation, confusion, restlessness, convulsions, coma, and urinary retention indicate atropine poisoning, requiring an immediate cessation of atropine administration.
Nicotinic Cholinergic Receptor Antagonists
These are also referred to as central anticholinergic agents, such as scopolamine, benactyzine, benztropine mesylate, and procyclidine. These agents primarily act on central muscarinic (M) and nicotinic (N) receptors but demonstrate weak effects on peripheral muscarinic (M) receptors.
New Anticholinergic Medications
Penehyclidine hydrochloride is a novel selective anticholinergic medication. It acts on both peripheral muscarinic receptors and central M and N receptors. It exhibits strong selectivity for M1 and M3 receptors and minimal effects on M2 receptors located in the heart, thereby not increasing heart rate or myocardial oxygen consumption. Compared with atropine, its anticholinergic effect is greater, its elimination half-life reaches 10.3 hours, and it requires a smaller effective dose while offering a prolonged duration of action. The choice of initial dose depends on the severity of poisoning and is delivered via intramuscular injection. The initial administration is combined with pralidoxime chloride.
For mild OPI poisoning, only cholinesterase reactivators are utilized. For moderate or severe poisoning, these are combined with cholinergic receptor antagonists. When combining the two drugs, the dose of the cholinergic receptor antagonist (e.g., atropine) is reduced to avoid toxicity.
Combination Preparations
Cholinergic receptor antagonists and cholinesterase reactivators can be formulated into combination preparations. Antidotal injections are available containing 3 mg of atropine, 3 mg of benactyzine, and 400 mg of pralidoxime chloride per vial. The initial dosage is administered via intramuscular injection as follows: 1/2–1 vial for mild poisoning, 1–2 vials for moderate poisoning, and 2–3 vials for severe poisoning. In addition, pralidoxime chloride is separately administered at doses of 0–0.5 g for mild poisoning, 0.5–1.0 g for moderate poisoning, and 1.0–1.5 g for severe poisoning.
For severe cases, the dosage of antidotes is gradually reduced after symptom relief. Once symptoms have nearly resolved and whole blood cholinesterase activity has risen to 50%–60% of normal, discontinuation and observation follow. Observation typically lasts 3–7 days before discharge.
Symptom Management
Patients with severe OPI poisoning often experience complications such as acidosis, hypokalemia, severe cardiac arrhythmias, and cerebral edema. When severe respiratory and circulatory failure is present, timely intervention is critical. If such complications are not managed promptly, the patient may succumb before antidotal therapy becomes effective.
Treatment of Intermediate Syndrome
Mechanical ventilation is initiated promptly. Pralidoxime chloride is administered intramuscularly at 1.0 g per dose, with the interval between doses adjusted based on the clinical condition, and treatment generally continues for 2–3 days. Symptomatic treatment is also actively undertaken.
Prevention
Raising awareness about OPI poisoning prevention and treatment among individuals involved in the production and use of OPIs is essential. Strict adherence to safety protocols and operating procedures is emphasized during the manufacture and processing of OPIs. Proper safety measures must be observed during the handling and application of pesticides. For individuals with chronic exposure, regular medical check-ups and monitoring of whole blood cholinesterase activity are recommended.