Rodenticides refer to chemical compounds capable of exterminating rodent species, such as rats and mice. There are more than ten types of rodenticides both domestically and internationally. Presently, rodenticides are widely used in rural and urban areas, with many of them exhibiting high toxicity to humans and animals upon ingestion. As a result, mass and sporadic poisoning incidents involving rodenticides have been frequently reported. Classifying rodenticides based on their speed of onset and toxicological action serves as a key reference for the effective treatment of rodenticide poisoning.
Classification of Poisoning
Classification Based on Speed of Onset
Acute Rodenticides
These cause death within 24 hours of ingestion and include agents such as tetramine (chemical name: tetramethylenedisulfotetramine) and fluoroacetamide.
Chronic Rodenticides
Death occurs several days after ingestion. Examples include anticoagulant rodenticides such as diphacinone sodium (diphacinone-Na) and warfarin.
Classification Based on Toxicological Action
Anticoagulant Rodenticides
First-generation high-toxicity anticoagulants: Examples include warfarin, coumafuryl, diphacinone sodium, and chlorophacinone.
Second-generation extremely toxic anticoagulants: Examples include brodifacoum and bromadiolone.
CNS-Stimulant Rodenticides
Examples include tetramine, fluoroacetamide, and sodium fluoroacetate.
Other Types of Rodenticides
These include:
- ANTU (increases capillary permeability);
- Pyrinuron (inhibits nicotinamide metabolism);
- Phosazetin (an organophosphate rodenticide);
- Zinc phosphide (an inorganic phosphorus agent);
- Crimidine (a vitamin B6 antagonist).
Etiology
Common causes of rodenticide poisoning include the following:
- Accidental ingestion or misuse of rodenticide-formulated baits.
- Intentional poisoning or homicidal poisoning.
- Secondary Poisoning: Rodenticides retained in the bodies of poisoned animals or plants in their original form may lead to poisoning when humans consume or utilize these contaminated animals or plants.
- Skin contact or inhalation during production or processing, leading to poisoning through dermal absorption or respiratory exposure.
Mechanisms of Toxicity and Principles of Treatment
Tetramine
Tetramine is the most common cause of fatal rodenticide poisoning, with a lethal dose for humans ranging from 5–12 mg (0.1–0.3 mg/kg) when ingested orally. It exhibits strong excitatory effects on the central nervous system, leading to intense convulsions following poisoning. Studies have indicated that the mechanism behind seizures involves tetramine antagonizing the inhibitory neurotransmitter gamma-aminobutyric acid (GABA) in the CNS. When the inhibitory action of GABA is blocked by tetramine, excessive excitation results, leading to convulsions. Tetramine’s severe toxicity and chemical stability also make secondary poisoning likely. No specific antidote is currently available.
Fluoroacetamide
Fluoroacetamide is an odorless and tasteless water-soluble white powder with an oral lethal dose for humans of 0.1–0.5 g. It can be absorbed via ingestion, inhalation, ocular exposure, or contact through open wounds. In the body, fluoroacetamide undergoes deamination to form fluoroacetate, which binds to coenzyme A. Under the action of oxaloacetate, fluoroacetyl-CoA forms fluorocitrate. While fluorocitrate is chemically similar to citrate, it is not metabolized by aconitase and instead inhibits this enzyme, preventing citrate metabolism and blocking the tricarboxylic acid (TCA) cycle, a phenomenon known as “lethal synthesis.”
The accumulation of citrate and the obstruction of pyruvate metabolism result in degeneration and necrosis of cardiac, cerebral, pulmonary, hepatic, and renal cells, leading to pulmonary and cerebral edema. Fluoroacetamide is also prone to causing secondary poisoning. Acetamide (a commercial antidote for fluoroacetamide poisoning) serves as its specific antidote. Its detoxification mechanism involves acetamide competing with fluoroacetamide for amidohydrolase, thereby reducing the conversion of fluoroacetamide into fluoroacetate. Additionally, acetamide provides acetyl groups for coenzyme A to form acetyl-CoA, which helps restore the tricarboxylic acid (TCA) cycle.
Brodifacoum and Bromadiolone
These rodenticides are among the most widely used globally and exhibit high toxicity to rodents while maintaining relatively high safety profiles for humans. Such rodenticides inhibit vitamin K epoxide reductase, thereby disrupting the liver’s utilization of vitamin K. This interference prevents gamma-carboxylation of vitamin K-dependent clotting factors (including factors II, VII, IX, and X), rendering them unable to bind calcium and thus inactive for coagulation, leading to prolonged clotting times. The breakdown product, benzylacetone, severely damages capillary endothelium, increasing vascular permeability and fragility, which can result in hemorrhages. Vitamin K1 is considered the specific antidote for poisoning caused by these compounds.
Zinc Phosphide
Zinc phosphide is commonly available in the form of powders, pellets, or tablets and is a low-cost, highly toxic rodenticide with a lethal dose of 40 mg/kg for humans. After ingestion, it reacts with gastric acids to release phosphine gas and zinc chloride. Phosphine gas inhibits cytochrome oxidase, causing cellular respiratory dysfunction, while zinc chloride exerts strong irritant and corrosive effects on the gastric mucosa, leading to gastric bleeding and ulceration. Inhalation of zinc phosphide can result in severe damage to cardiovascular, endocrine, hepatic, and renal functions, leading to multi-organ failure. Zinc phosphide reacts with copper sulfate to form insoluble copper phosphide, which precipitates, and potassium permanganate can convert zinc phosphide into non-toxic phosphoric anhydride. Therefore, for oral poisoning, copper sulfate solution and potassium permanganate solution can be used for detoxification.
Clinical Features and Diagnostic Points
Detailed information can be found in Table 1.
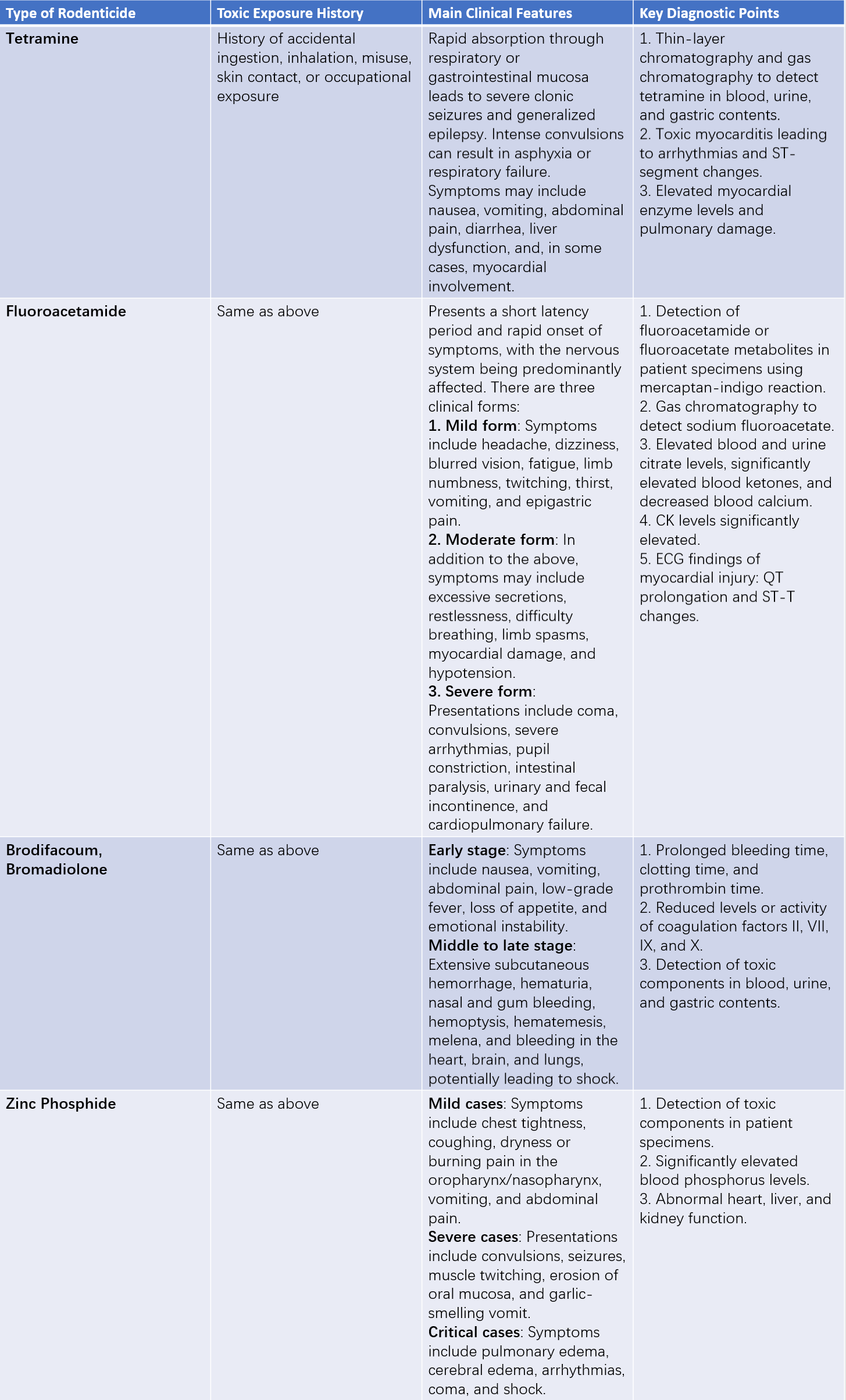
Table 1 Overview of the clinical features and diagnostic points of rodenticide poisoning
Treatment
Decontamination treatment should be performed as soon as possible following rodenticide poisoning. For cases involving dermal absorption or skin contamination, removal of contaminated clothing and thorough washing of the skin, hair, and nails in the affected area is recommended. For cases of oral poisoning, gastrointestinal decontamination should be carried out promptly and may include induced vomiting, gastric lavage, adsorption therapy, the use of laxatives, or whole bowel irrigation. Where a specific antidote exists for the toxin, it should be administered at the earliest opportunity. Monitoring of vital signs is essential, along with assessment of the patient’s airway, breathing, and circulatory status. There should be vigilance for airway obstruction risks caused by procedures such as gastric lavage, vomiting, or convulsions. Supportive treatment for organ function may be required in certain situations.
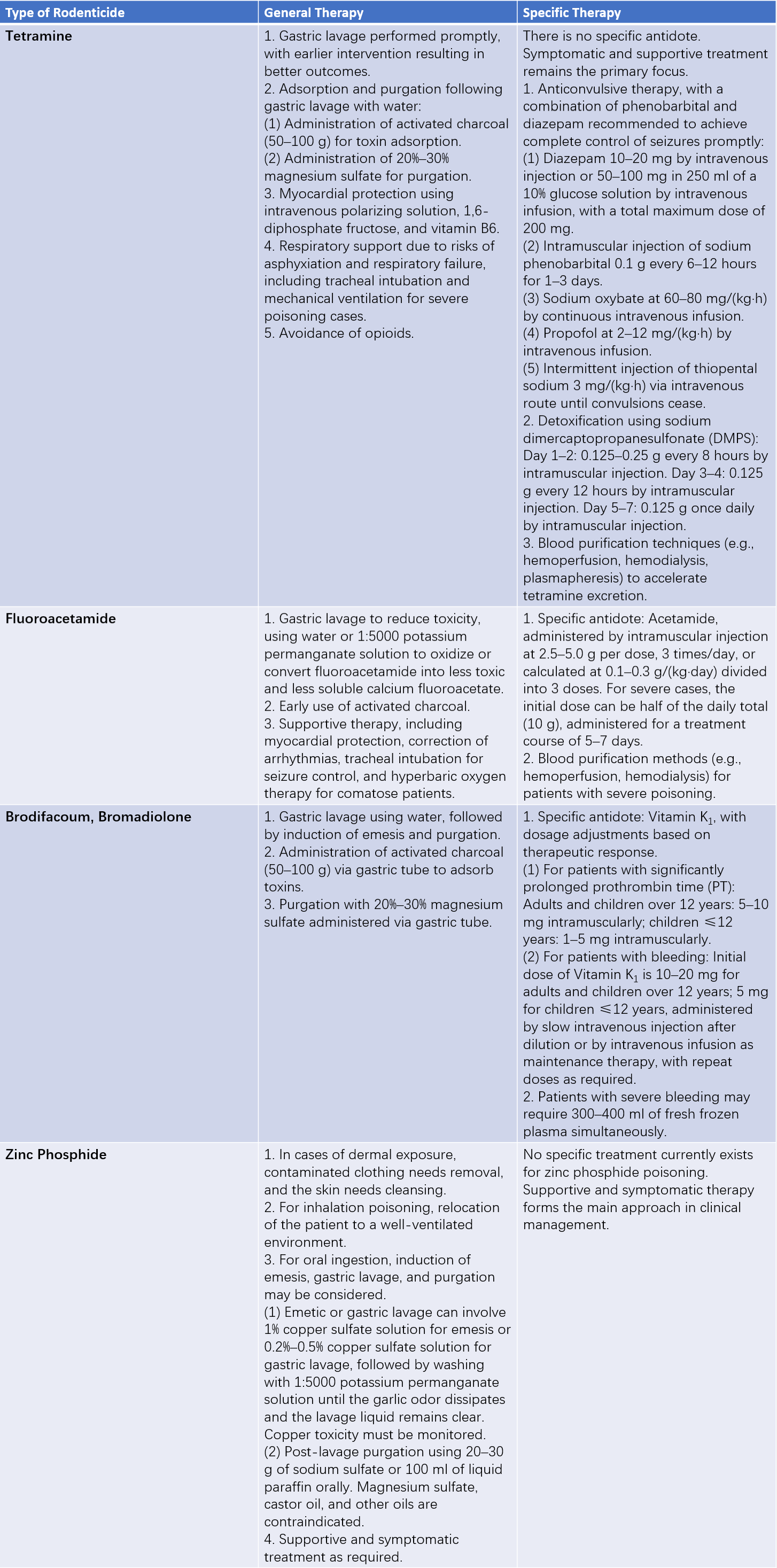
Table 2 Summary of treatment methods for rodenticide poisoning