Drugs (narcotics) refer to substances regulated by the state that include addictive narcotic analgesics and psychotropic drugs. These substances are characterized by drug dependence, harmfulness, and illegality. The term "drug" is relative: when used for medical purposes, they are considered medications, but in the case of misuse or abuse, they are categorized as drugs. Notably, the term does not include addictive substances found in tobacco or alcohol. Acute drug poisoning (acute narcotics poisoning) occurs when substances are abused, misused, or deliberately consumed in high doses within a short period, exceeding the body’s tolerance and producing clinical manifestations. Death in cases of acute drug poisoning often results from respiratory or circulatory failure, with accidental fatalities occasionally reported. Acute drug poisoning is primarily observed in individuals who abuse drugs. Currently, over 200 countries and regions worldwide face issues of drug abuse. In 2020, approximately 275 million people globally used drugs, with the most commonly abused substances including marijuana, amphetamines, heroin, cocaine, and ketamine.
Categories of Drugs
Drugs are divided into two main categories: narcotic (analgesic) drugs and psychotropic drugs. This section highlights some commonly encountered drugs.
Narcotic (Analgesic) Drugs
Opiates
Opium is a dried substance derived from the sap of unripe poppy capsules and exhibits strong analgesic, antitussive, antidiarrheal, sedative, and hypnotic effects. Opium contains more than 20 alkaloids, including morphine, codeine, thebaine, and papaverine. Thebaine serves as an antagonist to the effects of morphine and codeine but can be chemically modified to produce etorphine, a highly potent analgesic. Papaverine does not act on opioid receptors in the body. Opioid analgesics include natural opiate preparations, semi-synthetic opiates, and synthetic opioids, which act on opioid receptors in the body to provide analgesic effects.
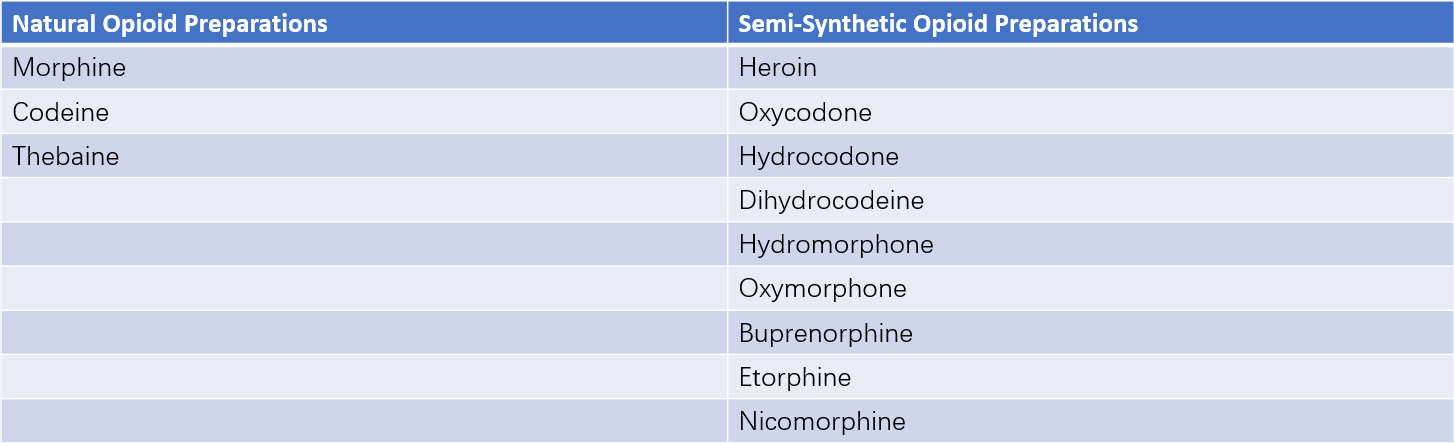
Table 1 Natural and semi-synthetic opioid preparations
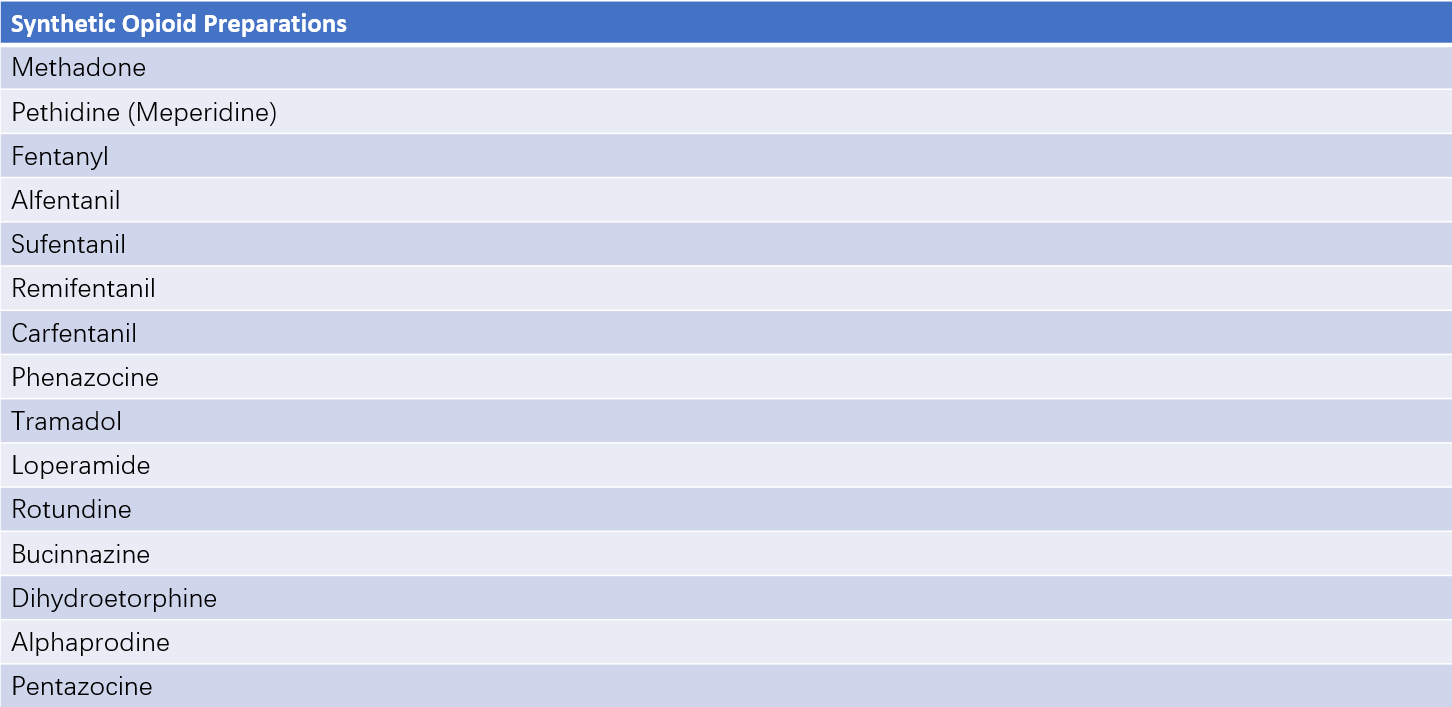
Table 2 Synthetic opioid preparations
Cocaine
This class includes substances such as cocaine, coca leaves, and coca paste. Cocaine (chemical name: benzoyl methyl ecgonine) is an alkaloid derived from coca leaves.
Cannabis
This category comprises marijuana leaves, cannabis resin, and cannabis oil, with psychoactive substances including delta-9-tetrahydrocannabinol (Δ9-THC), cannabidiol, cannabinol, and their respective acids.
Psychotropic Drugs
Central Nervous System Depressants
This includes sedative-hypnotic drugs and antianxiety medications. Poisoning associated with these substances is detailed under the chapter on sedative-hypnotic poisoning.
Central Stimulants
Commonly abused stimulants include amphetamine (AA) and its derivatives, such as methamphetamine (MA), 3,4-methylenedioxyamphetamine (MDA), and 3,4-methylenedioxymethamphetamine (MDMA).
Hallucinogens
This category includes substances like lysergide (LSD), phencyclidine (PCP), psilocybin, and mescaline. Ketamine, a derivative of PCP, is classified as a Category I psychotropic drug.
Causes of Poisoning
Most cases of drug poisoning result from abuse. Modes of abuse include oral ingestion, inhalation (e.g., nasal snorting, smoking, or "chasing"), injection (e.g., subcutaneous, intramuscular, intravenous, or intra-arterial), or mucosal rubbing (e.g., oral, nasal, or rectal application). Other causes include accidental ingestion, misuse, or deliberate high-dose use. Drug poisoning can also occur due to excessive medication use or frequent administration beyond the body’s tolerance for therapeutic reasons. Situations that increase the likelihood of poisoning among drug users include:
- Severe liver or kidney diseases.
- Severe pulmonary diseases.
- Gastric emptying delays.
- Severe hypothyroidism or adrenal cortical insufficiency.
- Combined ingestion of opioids with alcohol or sedative-hypnotic drugs.
- Elderly individuals with frail constitutions.
The majority of individuals experiencing poisoning due to drug abuse are adolescents.
Mechanism of Poisoning
Narcotic Drugs
Opioids
The onset time of toxic effects from opioid drugs varies depending on the route of administration. Effects can occur within 1–2 hours with oral ingestion, 10–15 minutes with nasal mucosal absorption, 10 minutes with intravenous injection, 30 minutes with intramuscular injection, or approximately 90 minutes with subcutaneous injection. The duration of action for opioids depends on the metabolic rate in the liver. Around 90% of the drug is excreted in urine as inactive metabolites, with a small portion excreted unchanged in urine or eliminated through bile and gastrointestinal contents in feces. Most of the drug is excreted within 24 hours, with urine tests showing almost no detectable amounts after 48 hours. Lipid-soluble opioids (such as morphine, heroin, propoxyphene, fentanyl, and buprenorphine) rapidly distribute to tissues after entering the bloodstream, including fetal tissues, and are stored in fatty tissues. Repeated administration of these substances can prolong their duration of action.
Morphine undergoes glucuronidation or N-demethylation in the liver to form N-demethyl morphine. Heroin has a low affinity for opioid receptors but is more lipophilic compared to morphine, enabling it to cross the blood-brain barrier more easily. Heroin has a plasma half-life of 3–9 minutes and is hydrolyzed by esterases into 6-monoacetylmorphine, which is metabolized into morphine within 45 minutes and exerts effects in the brain. Normeperidine, an active metabolite of meperidine (pethidine), exhibits strong neurotoxicity and may cause seizures.
Opioid receptors in the body include three main types, μ (μ1, μ2), κ, and δ, which are primarily located in pathways and regions associated with pain transmission, such as the periaqueductal gray, locus coeruleus, limbic system, and raphe magnus nucleus. These receptors are also found at sensory nerve endings, mast cells, and in the gastrointestinal tract. Genetic variations in opioid receptors may explain some individual differences in response to endogenous or exogenous opioids. Opioid receptors mediate the pharmacological effects of opioid drugs, including analgesia, sedation, respiratory depression, hallucinations, and euphoria.
Opioids are classified as receptor agonists or partial agonists (agonist/antagonist). Agonists primarily act on μ receptors and include drugs such as morphine, pethidine, methadone, fentanyl, and codeine. Partial agonists mainly act on κ receptors and antagonize μ receptors to varying degrees; these include drugs like pentazocine, buprenorphine, and butorphanol. When opioids activate central nervous system opioid receptors, they produce effects such as analgesia, sedation, respiratory depression, hallucinations, and euphoria. Prolonged use can lead to drug dependence. Opioid dependence and withdrawal syndromes are believed to share similar mechanisms. Specifically, opioids bind to opioid receptors, inhibiting the production of endogenous opioids (endorphins). When opioid use stops, the body cannot immediately compensate by producing sufficient endorphins, leading to withdrawal symptoms.
For adults, the lethal dose of orally ingested opium is 2–5 g. The acute toxic dose of morphine for intramuscular injection is 60 mg, with a lethal dose of approximately 250–300 mg. First-time users may experience toxicity with an oral dose of 120 mg of opium or an intramuscular dose of more than 30 mg of morphine. However, dependent individuals can tolerate up to 5 g of morphine sulfate intravenously within 24 hours without showing signs of poisoning. The toxic dose of codeine is 200 mg, with a lethal dose of 800 mg. For heroin, a toxic dose is 50–100 mg, and the lethal dose ranges from 750–1,200 mg. Meperidine has a lethal dose of approximately 1.0 g.
Cocaine
Cocaine is a lipid-soluble compound and an ancient local anesthetic with strong central stimulant effects. After absorption via mucous membranes, it quickly enters the bloodstream and readily crosses the blood-brain barrier. Cocaine has both central nervous system stimulant and sympathomimetic actions, primarily due to the inactivation of serotonin (5-HT) and dopamine transporters in the brain. Abusers exhibit strong psychological dependence, and repeated high-dose use can lead to physiological dependence. Withdrawal symptoms may occur upon cessation, although its addictive potential is lower than morphine and heroin. Acute toxic doses vary widely among individuals, with a toxic dose of 20 mg and a lethal dose of 1,200 mg. In some cases, even a dose of 70 mg of pure cocaine can cause immediate death in a 70 kg adult.
Acute cocaine poisoning increases the release of dopamine, epinephrine, norepinephrine, and 5-HT. These neurotransmitters act on various receptor subtypes, leading to multiple effects. For example, epinephrine and norepinephrine can increase heart rate, enhance myocardial contractility, and elevate blood pressure. Cocaine blocks sodium channels in cardiac myocytes, similar to class Ia antiarrhythmic drugs, potentially causing cardiac conduction abnormalities during acute poisoning. At high doses, cocaine suppresses the respiratory center and may result in cardiac arrest when administered intravenously.
Cannabis
The mechanism of action of cannabis remains unclear. Acute poisoning produces effects similar to alcohol, affecting the nervous, psychiatric, respiratory, and circulatory systems. Long-term use leads to psychological dependence but does not result in physiological dependence.
Psychotropic Drugs
Amphetamines
Amphetamine (AA) is a non-catecholamine sympathomimetic amine with low molecular weight that easily crosses the blood-brain barrier after absorption. Its primary mechanism of action involves promoting the release of catecholamine neurotransmitters (dopamine and norepinephrine) in the brain while reducing levels of the inhibitory neurotransmitter serotonin (5-HT), resulting in neural excitation and euphoria. The toxic dose varies significantly among individuals, with the lethal oral dose for healthy adults ranging from 20–25 mg/kg. Methamphetamine (MA) is approximately twice as toxic as amphetamine. Acute poisoning can occur in minutes after intravenous injection of 10 mg, with toxicity sometimes being observed at doses as low as 2 mg. Chronic users may require intravenous doses of 30–50 mg, while tolerant individuals may not exhibit toxicity even with injections exceeding 1,000 mg.
Ketamine
Ketamine is a newer, non-barbiturate intravenous anesthetic that exerts its anesthetic effects primarily in brain tissue upon administration. Most of the drug undergoes hepatic metabolism, converting into norketamine, which is further metabolized into dehydronorketamine, an active metabolite. In the liver, it may also conjugate with glucuronic acid. A small amount of unmetabolized ketamine and most of its metabolites are excreted through the kidneys. Ketamine acts as a specific antagonist of N-methyl-D-aspartate (NMDA) receptors, blocking the transmission of pain impulses to the thalamo-neocortical system, thus providing analgesic effects. It stimulates the brainstem and limbic system, causing dissociation between consciousness and sensory perception. Ketamine excites the sympathetic nervous system, though high doses administered rapidly may inhibit respiration. It also exhibits antagonistic activity at μ-opioid receptors and agonistic effects at κ-opioid receptors.
Diagnosis
Diagnosis is often based on a history of substance abuse, clinical symptoms, laboratory tests, and detoxification test responses. Diagnosis can be more challenging in cases involving multiple drug intoxications.
History of Use or Inhalation
For narcotic drugs used for therapeutic purposes, the patient's history is usually clear. However, in cases of abuse or overdose, obtaining an accurate history can be difficult. Examination may reveal signs of drug use, such as nasal or oral ulcers and perforations from inhalation or burn scars, or injection marks on the skin from intravenous use. Psychotropic drug abuse is more commonly observed among young individuals frequenting certain social or entertainment venues.
Clinical Manifestations of Acute Poisoning
Narcotic Drugs
Opioid Toxicity
A typical triad of coma, respiratory depression, and pinpoint pupils (miosis) is often observed. Morphine toxicity is characterized by this triad along with cyanosis and hypotension. Heroin toxicity may cause non-cardiogenic pulmonary edema. Pethidine (meperidine) can trigger seizures, convulsions, delirium, tachycardia, and dilated pupils. Fentanyl is commonly associated with chest wall rigidity. Methadone toxicity has been linked to blindness and lower-limb paralysis. Most acute opioid poisonings result in death within 12 hours due to respiratory failure; however, patients surviving beyond 48 hours generally have a better prognosis. Comatose patients with opioid toxicity may also develop rhabdomyolysis, myoglobinuria, renal failure, and compartment syndrome.
Cocaine Toxicity
This is less commonly observed. Severe acute toxicity presents with symptoms such as intense itching, limb tremors, muscle spasms, grand mal seizures, hyperthermia, elevated blood pressure, dilated pupils, increased heart rate, rapid breathing, and hyperreflexia.
Cannabis Toxicity
Acute poisoning can occur after a single large dose, manifesting as psychological and behavioral abnormalities, including hyperthermic delirium, panic, agitation, altered consciousness, or coma. Some individuals may experience transient depressive states, feelings of despair, or suicidal ideation. Examination may reveal conjunctival hyperemia, tachycardia, and elevated blood pressure.
Psychotropic Drugs
Amphetamine Toxicity
Symptoms include psychic excitement, hyperactivity, anxiety, tension, hallucinations, and disorientation. Severe cases may present with sweating, facial flushing, dilated pupils, elevated blood pressure, tachycardia or ventricular arrhythmias, increased respiration, hyperthermia, tremors, muscle spasms, convulsions, or coma. Hypertension may lead to intracranial hemorrhages. Common causes of death include disseminated intravascular coagulation (DIC), circulatory failure, or liver and kidney failure.
Ketamine Toxicity
Symptoms primarily involve neurological and psychiatric effects, such as confusion, slurred speech, hallucinations, hyperthermia, delirium, tremors, and catatonia.
Laboratory Tests
Toxicology Screening
For oral poisoning cases, analysis of gastric contents, vomitus, or samples of urine and blood is conducted to perform qualitative toxicology screening. Measuring blood drug concentrations, when feasible, further aids in diagnosis.
Urine Testing
In suspected heroin poisoning, urine should be collected in 4 hours for toxicology screening. High-performance liquid chromatography (HPLC) can detect amphetamine (AA) and its metabolites in urine. Ketamine and its metabolites can also be identified in urine to support the diagnosis.
Blood Testing
Morphine
Therapeutic plasma concentration ranges from 0.01–0.07 mg/L, with toxic concentrations ranging from 0.1–1.0 mg/L and lethal concentrations exceeding 4.0 mg/L.
Methadone
Therapeutic plasma concentration ranges from 0.48–0.85 mg/L, with toxic concentrations at 2.0 mg/L and lethal concentrations reaching 74.0 mg/L.
Amphetamine
Toxic plasma concentration is 0.5 mg/L, while lethal concentrations exceed 2.0 mg/L.
Other Examinations
Arterial Blood Gas Analysis
Patients with severe narcotic poisoning often exhibit hypoxemia and respiratory acidosis.
Blood Biochemistry Tests
Assessments include blood glucose, electrolytes, and liver and renal function tests.
Differential Diagnosis
In cases of opioid poisoning accompanied by delirium, the involvement of other psychotropic drugs or coexisting brain disorders should be considered. Patients with pinpoint pupils should be differentially diagnosed for sedative-hypnotic drugs, phenothiazine, opium alkaloids (OPI), clonidine poisoning, or pontine hemorrhage. Since heroin is often adulterated with other substances (e.g., quinine, caffeine, or diazepam), atypical poisoning presentations may be influenced by the adulterants. Patients experiencing opioid withdrawal syndrome typically do not exhibit cognitive changes; thus, any cognitive changes should prompt investigation of other potential causes.
Diagnostic Therapy
Administering antidotes specific to the suspected drug and observing the therapeutic response can assist in diagnosis. For example, in suspected morphine poisoning, intravenous naloxone rapidly relieves symptoms.
Treatment
Resuscitation and Support Therapy
In drug poisoning cases complicated by respiratory or circulatory failure, resuscitation measures take precedence.
Respiratory Support
For respiratory failure, appropriate measures include:
- Maintaining airway patency, with endotracheal intubation or tracheostomy performed if necessary.
- Using atropine to stimulate the respiratory center or employing central nervous system stimulants such as caffeine and sodium benzoate or nicetamide. Drugs like strychnine or tubocurarine must be avoided, as they may synergize with morphine to cause or exacerbate convulsions.
- Implementing mechanical ventilation with positive end-expiratory pressure (PEEP), which effectively treats non-cardiogenic pulmonary edema induced by heroin or methadone poisoning. High-concentration oxygen, vasodilators, and loop diuretics can be administered, while theophylline should be avoided.
Circulatory Support
For patients with hypotension, elevating the lower body, administering intravenous fluids, and using vasopressors when necessary are conducted. In arrhythmias induced by propoxyphene, class Ia antiarrhythmic drugs should be avoided. Ventricular arrhythmias caused by cocaine poisoning can be treated with labetalol or phenytoin.
Correction of Metabolic Disorders
Hypoglycemia, acidosis, and electrolyte imbalances should be corrected as required.
Toxin Elimination
Emesis
Use of apomorphine for inducing vomiting in patients with clear consciousness is contraindicated to prevent aggravated toxicity.
Gastric Lavage
If a lethal dose of the drug is consumed, gastric lavage is performed within 1 hour. Initially, a 0.02%–0.05% potassium permanganate solution may be used, followed by 50% magnesium sulfate for purgation.
Activated Charcoal Adsorption
Activated charcoal suspension is employed to adsorb unabsorbed toxins. In cases of propoxyphene overdose or poisoning, repeated doses of activated charcoal may be particularly effective due to the enterohepatic circulation of the drug.
Antidotes
Naloxone
Naloxone can be administered intravenously, intramuscularly, subcutaneously, or intratracheally. For opioid poisoning, a 2 mg dose is given intravenously. In cases of opioid dependence-related poisoning, doses can be repeated every 3–10 minutes, while for non-dependent cases, doses may be repeated every 2–3 minutes. If the cumulative dose reaches 15–20 mg without effect, other conditions should be considered, such as poisoning by non-opioid substances (e.g., barbiturates), head injury, central nervous system disorders, or severe cerebral hypoxia. For poisoning involving long half-life opioids (e.g., methadone) or potent opioids (e.g., fentanyl), naloxone should be administered through intravenous infusion. Naloxone exhibits antagonistic effects against morphine that are 30 times stronger than those of nalorphine. A 1 mg dose of naloxone can counteract the effects of 25 mg of intravenous heroin. While effective for fentanyl-induced muscle rigidity, naloxone is ineffective in treating seizures caused by pethidine poisoning and non-cardiogenic pulmonary edema induced by heroin or methadone poisoning.
Nalmefene
Nalmefene is more effective than naloxone in treating morphine poisoning. It is administered intravenously at doses of 0.1–0.5 mg, titrating the dose gradually every 2–3 minutes, with a maximum dose of 1.6 mg per occurrence.
Nalorphine
Nalorphine shares a similar chemical structure with morphine and exerts direct antagonistic effects against morphine. It is used to treat acute poisoning caused by morphine, its derivatives, or other analgesics. A dose of 5–10 mg is administered intramuscularly or intravenously, with repeat doses every 20 minutes if needed, not exceeding a total of 40 mg.
Levallorphan
Levallorphan is an opioid antagonist that reverses respiratory depression caused by opioid poisoning. It cannot reverse, and may even worsen, respiratory depression caused by non-opioid central depressants (e.g., ethanol). An initial intravenous dose of 1–2 mg is given, followed by 0.5 mg doses at intervals of 5–15 minutes, for a total of 1–2 additional administrations as needed.
Naltrexone
Naltrexone shares structural similarity with naloxone but demonstrates stronger affinity for opioid receptors, with 3.6 times the affinity for μ receptors compared to naloxone. Its potency is twice that of naloxone and 17 times that of nalorphine. Naltrexone is rapidly absorbed orally, with a half-life of 4–10 hours, and its effects last for 24 hours. Both its metabolites and unchanged drug are excreted primarily via the kidneys. Naltrexone is used for opioid detoxification and relapse prevention, with a recommended dosage of 50 mg per day.
Symptomatic Treatment
Hyperthermia
Physical cooling methods such as alcohol sponge baths, ice packs, or ice caps are employed.
Seizures
Seizures caused by psychotropic drug poisoning are managed with thiopental sodium or diazepam.
Chest Wall Muscle Rigidity
Muscle relaxants are administered in cases of severe muscle rigidity.
Severe Malnutrition
Nutritional support therapy is recommended for patients with significant malnutrition.
Prevention
Management of anesthetics and psychotropic drugs should be strengthened, with designated personnel assigned for storage and oversight.
Indications, dosages, and durations of drug use should be strictly adhered to in order to avoid misuse and overuse.
Use in patients with liver, kidney, or lung dysfunction should be avoided, and dosage reductions should be considered for critically ill, elderly, or frail individuals.
Co-administration with other respiratory depressant drugs should be avoided when used therapeutically.
Patients treated successfully with naloxone for opioid poisoning should remain under observation to monitor for recurrence of opioid toxicity after naloxone's effects subside.